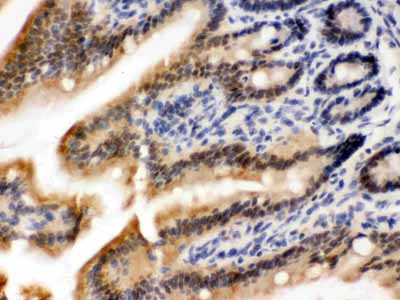
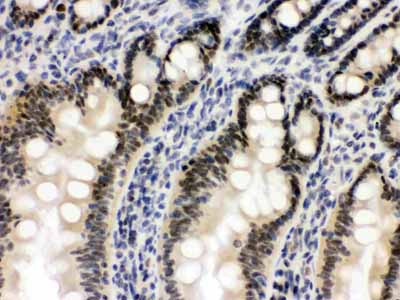
Anti-OGT Picoband Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC-P |
|---|---|
| Primary Accession | O15294 |
| Host | Rabbit |
| Reactivity | Human, Mouse, Rat |
| Clonality | Polyclonal |
| Format | Lyophilized |
| Description | Rabbit IgG polyclonal antibody for UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit(OGT) detection. Tested with WB, IHC-P in Human;Mouse;Rat. |
| Reconstitution | Add 0.2ml of distilled water will yield a concentration of 500ug/ml. |
| Gene ID | 8473 |
|---|---|
| Other Names | UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit, 2.4.1.255, O-GlcNAc transferase subunit p110, O-linked N-acetylglucosamine transferase 110 kDa subunit, OGT, OGT |
| Calculated MW | 116925 MW KDa |
| Application Details | Immunohistochemistry(Paraffin-embedded Section), 0.5-1 µg/ml, Mouse, Rat, Human, By Heat Western blot, 0.1-0.5 µg/ml, Human, Mouse, Rat |
| Subcellular Localization | Isoform 2: Mitochondrion. Membrane. Associates with the mitochondrial inner membrane. |
| Tissue Specificity | Highly expressed in pancreas and to a lesser extent in skeletal muscle, heart, brain and placenta. Present in trace amounts in lung and liver. . |
| Protein Name | UDP-N-acetylglucosamine--peptide N-acetylglucosaminyltransferase 110 kDa subunit |
| Contents | Each vial contains 5mg BSA, 0.9mg NaCl, 0.2mg Na2HPO4, 0.05mg NaN3. |
| Immunogen | A synthetic peptide corresponding to a sequence at the C-terminus of human OGT (1008-1046aa NTKQYTMELERLYLQMWEHYAAGNKPDHMIKPVEVTESA), identical to the related mouse and rat sequences. |
| Purification | Immunogen affinity purified. |
| Cross Reactivity | No cross reactivity with other proteins. |
| Storage | At -20˚C for one year. After r˚Constitution, at 4˚C for one month. It˚Can also be aliquotted and stored frozen at -20˚C for a longer time.Avoid repeated freezing and thawing. |
| Name | OGT {ECO:0000303|PubMed:11773972, ECO:0000312|HGNC:HGNC:8127} |
|---|---|
| Function | Catalyzes the transfer of a single N-acetylglucosamine from UDP-GlcNAc to a serine or threonine residue in cytoplasmic and nuclear proteins resulting in their modification with a beta-linked N- acetylglucosamine (O-GlcNAc) (PubMed:12150998, PubMed:19451179, PubMed:20018868, PubMed:26678539, PubMed:26369908, PubMed:23103939, PubMed:21240259, PubMed:21285374, PubMed:27713473, PubMed:15361863, PubMed:37541260, PubMed:26237509). Glycosylates a large and diverse number of proteins including histone H2B, AKT1, AMPK, ATG4B, CAPRIN1, EZH2, FNIP1, KRT7, LMNA, LMNB1, LMNB2, RPTOR, HOXA1, PFKL, KMT2E/MLL5, MAPT/TAU, TET2, RBL2, RET, NOD2 and HCFC1 (PubMed:19451179, PubMed:20200153, PubMed:21285374, PubMed:22923583, PubMed:23353889, PubMed:24474760, PubMed:26678539, PubMed:26369908, PubMed:27527864, PubMed:30699359, PubMed:34074792, PubMed:34667079, PubMed:37541260, PubMed:26237509). Can regulate their cellular processes via cross-talk between glycosylation and phosphorylation or by affecting proteolytic processing (PubMed:21285374). Involved in insulin resistance in muscle and adipocyte cells via glycosylating insulin signaling components and inhibiting the 'Thr-308' phosphorylation of AKT1, enhancing IRS1 phosphorylation and attenuating insulin signaling (By similarity). Involved in glycolysis regulation by mediating glycosylation of 6- phosphofructokinase PFKL, inhibiting its activity (PubMed:22923583). Plays a key role in chromatin structure by mediating O-GlcNAcylation of 'Ser-112' of histone H2B: recruited to CpG-rich transcription start sites of active genes via its interaction with TET proteins (TET1, TET2 or TET3) (PubMed:22121020, PubMed:23353889). As part of the NSL complex indirectly involved in acetylation of nucleosomal histone H4 on several lysine residues (PubMed:20018852). O-GlcNAcylation of 'Ser-75' of EZH2 increases its stability, and facilitating the formation of H3K27me3 by the PRC2/EED-EZH2 complex (PubMed:24474760). Stabilizes KMT2E/MLL5 by mediating its glycosylation, thereby preventing KMT2E/MLL5 ubiquitination (PubMed:26678539). Regulates circadian oscillation of the clock genes and glucose homeostasis in the liver (By similarity). Stabilizes clock proteins BMAL1 and CLOCK through O-glycosylation, which prevents their ubiquitination and subsequent degradation (By similarity). Promotes the CLOCK-BMAL1-mediated transcription of genes in the negative loop of the circadian clock such as PER1/2 and CRY1/2. O-glycosylates HCFC1 and regulates its proteolytic processing and transcriptional activity (PubMed:21285374, PubMed:28584052, PubMed:28302723). Component of a THAP1/THAP3-HCFC1-OGT complex that is required for the regulation of the transcriptional activity of RRM1 (PubMed:20200153). Regulates mitochondrial motility in neurons by mediating glycosylation of TRAK1 (By similarity). Promotes autophagy by mediating O-glycosylation of ATG4B (PubMed:27527864). Acts as a regulator of mTORC1 signaling by mediating O-glycosylation of RPTOR and FNIP1: O-GlcNAcylation of RPTOR in response to glucose sufficiency promotes activation of the mTORC1 complex (PubMed:30699359, PubMed:37541260). |
| Cellular Location | Nucleus. Cytoplasm. Note=Predominantly localizes to the nucleus (PubMed:26678539). Translocates into the nucleus via association with importin KPNA1 (PubMed:27713473) [Isoform 3]: Cytoplasm. Nucleus. Cell membrane {ECO:0000250|UniProtKB:P56558}. Mitochondrion membrane {ECO:0000250|UniProtKB:P56558}. Cell projection {ECO:0000250|UniProtKB:P56558}. Note=Mostly in the nucleus. Retained in the nucleus via interaction with HCFC1 (PubMed:21285374). After insulin induction, translocated from the nucleus to the cell membrane via phosphatidylinositide binding. Colocalizes with AKT1 at the plasma membrane. TRAK1 recruits this protein to mitochondria. In the absence of TRAK1, localizes in cytosol and nucleus (By similarity) {ECO:0000250|UniProtKB:P56558, ECO:0000269|PubMed:21285374} |
| Tissue Location | Highly expressed in pancreas and to a lesser extent in skeletal muscle, heart, brain and placenta. Present in trace amounts in lung and liver. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
O-linked N-acetylglucosamine (O-GlcNAc) transferase (OGT) is an enzyme that in humans is encoded by the OGT gene. This gene encodes a glycosyltransferase that catalyzes the addition of a single N-acetylglucosamine in O-glycosidic linkage to serine or threonine residues. Since both phosphorylation and glycosylation compete for similar serine or threonine residues, the two processes may compete for sites, or they may alter the substrate specificity of nearby sites by steric or electrostatic effects. The protein contains multiple tetratricopeptide repeats that are required for optimal recognition of substrates. Alternatively spliced transcript variants encoding distinct isoforms have been found for this gene.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.