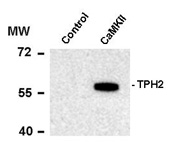
Phospho-Ser19 Tryptophan Hydroxylase 2 Antibody
Affinity purified rabbit polyclonal antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB |
|---|---|
| Primary Accession | Q8CGU9 |
| Reactivity | Rat |
| Predicted | Bovine, Mouse, Zebrafish |
| Host | Rabbit |
| Clonality | polyclonal |
| Calculated MW | 55 KDa |
| Gene ID | 317675 |
|---|---|
| Gene Name | TPH2 |
| Other Names | Tryptophan 5-hydroxylase 2, Neuronal tryptophan hydroxylase, Tryptophan 5-monooxygenase 2, Tph2, Ntph |
| Target/Specificity | Synthetic phospho-peptide corresponding to amino acid residues surrounding Ser19 conjugated to KLH. |
| Dilution | WB~~ 1:1000 |
| Format | Prepared from rabbit serum by affinity purification via sequential chromatography on phospho- and dephosphopeptide affinity columns. |
| Antibody Specificity | Specific for the ~55k tryptophan hydroxylase protein phosphorylated at Ser19. |
| Storage | Maintain refrigerated at 2-8°C for up to 6 months. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | Phospho-Ser19 Tryptophan Hydroxylase 2 Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Shipping | Blue Ice |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Tryptophan hydroxylase (TPH) catalyzes the 5-hydroxylation of tryptophan, which is the first step in the biosynthesis of indoleamines (serotonin and melatonin) (Martinez et al., 2001). In mammals, serotonin biosynthesis occurs predominantly in neurons which originate in the Raphe nuclei of the brain, and melatonin synthesis takes place within the pineal gland. Although TPH catalyzes the same reaction within the Raphe nuclei and the pineal gland, TPH activity is rate-limiting for serotonin but not melatonin biosynthesis. Serotonin functions mainly as a neurotransmitter, whereas melatonin is the principal hormone secreted by the pineal gland. The activity of TPH is enhanced by phosphorylation by cAMP-dependent protein kinase (PKA) and Ca2+/calmodulin kinase II (CaM K II) (Jiang et al., 2000; Johansen et al., 1996). CaM K II phosphorylates Ser19 which lies within the regulatory domain of TPH2 (McKinney et al., 2005).
References
Jiang GC, Yohrling GJ, Schmitt JD, Vrana KE (2000) Identification of substrate orienting and phosphorylation sites within tryptophan hydroxylase using homology-based molecular modeling. J Mol Biol 302:1005-1017.
Johansen PA, Jennings I, Cotton RG, Kuhn DM (1996) Phosphorylation and activation of tryptophan hydroxylase by exogenous protein kinase A. J Neurochem 66:817-823.
Martinez A, Knappskog PM, Haavik J (2001) Structural approach into human tryptophan hydroxylase and its implications for the regulation of serotonin biosynthesis. Curr Med Chem 8:1077-1091.
McKinney J, Knappskog PM, Haavik J (2005) Different properties of the central and peripheral forms of human tryptophan hydrpxylase. J Neurochem 92(2):311-20.
Kuhn DM, Sakowski SA, Geddes TJ, Wilkerson C, Haycock JW (2007) Phosphorylation and activation of tryptophan hydroxylase 2: identification of serine-19 as the substrate site for calcium, calmodulin-dependent protein kinase II. J Neurochem 103(4):1567-73.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.