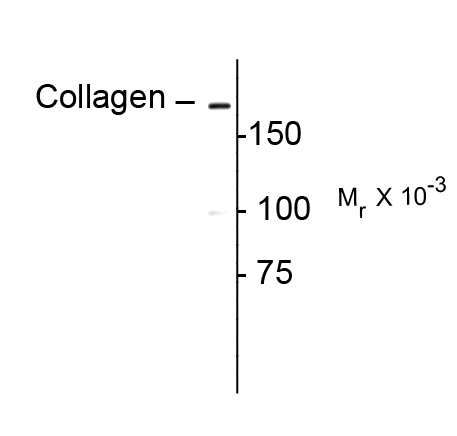Collagen I α1 Propeptide Sequence Antibody
Rabbit polyclonal antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC |
|---|---|
| Primary Accession | P02452 |
| Reactivity | Human, Mouse |
| Host | Rabbit |
| Clonality | polyclonal |
| Calculated MW | 180 KDa |
| Gene ID | 1277 |
|---|---|
| Gene Name | COL1A1 |
| Other Names | Collagen alpha-1(I) chain, Alpha-1 type I collagen, COL1A1 |
| Target/Specificity | Synthetic peptide corresponding to amino acid residues specific to the collagen 1, alpha 1 propeptide conjugated to KLH. |
| Dilution | WB~~ 1:1000 IHC~~ 1:100 |
| Format | Affinity purified |
| Antibody Specificity | Specific for the propeptide portion of the ~180 kDa collagen I α1polypeptide in human lung fibroblast extract. The antibody also works well forimmunohistochemistry on paraformaldehyde-fixed sections with a simple antigen-retrievalprotocol (incubate slides for 20 minutes at 90º C in 10 mM sodium citrate (pH 6.0)/ 0.1 %Tween-20). Note that in paraffin sections of formaldehyde-fixed fibrotic mouse lung tissue, theantibody recognizes collagen I molecules that are still associated with the cells in which theywere synthesized. |
| Storage | Maintain refrigerated at 2-8°C for up to 6 months. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | Collagen I α1 Propeptide Sequence Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Shipping | Blue Ice |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Collagen is an extracellular matrix protein that serves as a scaffold defining the shape and mechanical properties of many tissues and organs including skin, tendon, artery walls, fibrocartilage, bone and teeth. Type 1 collagen is the must abundant protein in mammals. Collagens are synthesized with N-terminal and C-terminal propeptides that are cleaved during maturation and secretion. After cleavage of the propeptides, the most N-terminal and C-terminal remaining sequences are known as telopeptides. Mutations in the collagen 1, alpha 1 gene (COL1A1) are known to cause osteogenesis imperfecta (aka brittle bone disease) (Byers 1989). Furthermore, mutations found in the fist 90 residues of the helical region of alpha 1 collagen have been implicated in the prevention or delayed removal of the procollagen N-propeptide leading to a combined osteogenesis imperfecta and Ehlers-Danlos syndrome (EDS) phenotype (Cabral et al., 2005).
References
Byers PH (1989) Inherited disorders of collagen gene structure and expression. Am J Med
Genet. 34(1):72-80.
Cabral WA, Makareeva E, Colige A, Letocha AD, Ty JM, Yeowell HN, Pals G, Leikin S, Marini
JC. (2005) Mutations near amino end of alpha1(I) collagen cause combined osteogenesis
imperfecta/Ehlers-Danlos syndrome by interference with N-propeptide processing. J Biol Chem.
2005 May 13;280(19):19259-69.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.