CDC2 Antibody
Purified Mouse Monoclonal Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
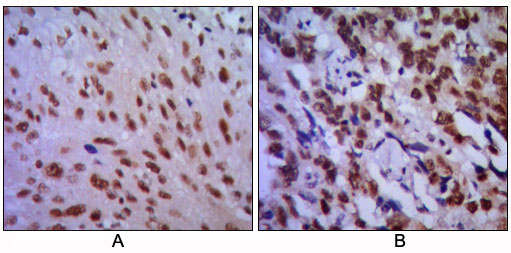
| WB, IHC, E |
|---|---|
| Primary Accession | P06493 |
| Reactivity | Human |
| Host | Mouse |
| Clonality | Monoclonal |
| Clone Names | 8C5A6 |
| Isotype | IgG1 |
| Calculated MW | 34kDa |
| Description | The cell division control protein cdc2, also known as cyclin-dependent kinase 1 (Cdk1) or p34/cdk1, plays a key role in the control of the eukaryotic cell cycle, where it is required for entry into S-phase and mitosis. Cdc2 exists as a complex with both cyclin A and cyclin B. The best characterized of these associations is the Cdc2 p34 cyclin B complex, which is required for the G2 to M phase transition. Activation of Cdc2 is controlled at several steps including cyclin binding and phosphorylation of threonine 161. However, the critical regulatory step in activating cdc2 during progression into mitosis appears to be dephosphorylation of Tyr15 and Tyr14. Phosphorylation at Tyr15 and inhibition of Cdc2 is carried out by WEE1 and MIK protein kinases while Tyr15 dephosphorylation and activation of Cdc2 is carried out by the cdc25 phosphatase. The isoform CDC2deltaT is found in breast cancer tissues. Furthermore, cdc2/Cdk1 is a key mediator of neuronal cell death in brain development and degeneration. |
| Immunogen | Purified recombinant fragment of CDC2 expressed in E. Coli. |
| Formulation | Ascitic fluid containing 0.03% sodium azide. |
| Gene ID | 983 |
|---|---|
| Other Names | Cyclin-dependent kinase 1, CDK1, 2.7.11.22, 2.7.11.23, Cell division control protein 2 homolog, Cell division protein kinase 1, p34 protein kinase, CDK1, CDC2, CDC28A, CDKN1, P34CDC2 |
| Dilution | WB~~1/500 - 1/2000 IHC~~1/200 - 1/1000 |
| Storage | Maintain refrigerated at 2-8°C for up to 6 months. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | CDC2 Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | CDK1 |
|---|---|
| Synonyms | CDC2, CDC28A, CDKN1, P34CDC2 |
| Function | Plays a key role in the control of the eukaryotic cell cycle by modulating the centrosome cycle as well as mitotic onset; promotes G2-M transition via association with multiple interphase cyclins (PubMed:2344612, PubMed:2188730, PubMed:16407259, PubMed:17459720, PubMed:16933150, PubMed:18356527, PubMed:19509060, PubMed:20171170, PubMed:19917720, PubMed:20937773, PubMed:20935635, PubMed:21063390, PubMed:23355470, PubMed:23601106, PubMed:23602554, PubMed:25556658, PubMed:26829474, PubMed:30139873, PubMed:30704899). Phosphorylates PARVA/actopaxin, APC, AMPH, APC, BARD1, Bcl-xL/BCL2L1, BRCA2, CALD1, CASP8, CDC7, CDC20, CDC25A, CDC25C, CC2D1A, CENPA, CSNK2 proteins/CKII, FZR1/CDH1, CDK7, CEBPB, CHAMP1, DMD/dystrophin, EEF1 proteins/EF-1, EZH2, KIF11/EG5, EGFR, FANCG, FOS, GFAP, GOLGA2/GM130, GRASP1, UBE2A/hHR6A, HIST1H1 proteins/histone H1, HMGA1, HIVEP3/KRC, KAT5, LMNA, LMNB, LBR, LATS1, MAP1B, MAP4, MARCKS, MCM2, MCM4, MKLP1, MLST8, MYB, NEFH, NFIC, NPC/nuclear pore complex, PITPNM1/NIR2, NPM1, NCL, NUCKS1, NPM1/numatrin, ORC1, PRKAR2A, EEF1E1/p18, EIF3F/p47, p53/TP53, NONO/p54NRB, PAPOLA, PLEC/plectin, RB1, TPPP, UL40/R2, RAB4A, RAP1GAP, RCC1, RPS6KB1/S6K1, KHDRBS1/SAM68, ESPL1, SKI, BIRC5/survivin, STIP1, TEX14, beta-tubulins, MAPT/TAU, NEDD1, VIM/vimentin, TK1, FOXO1, RUNX1/AML1, SAMHD1, SIRT2, CGAS and RUNX2 (PubMed:2344612, PubMed:2188730, PubMed:16407259, PubMed:17459720, PubMed:16933150, PubMed:18356527, PubMed:19509060, PubMed:20171170, PubMed:19917720, PubMed:20937773, PubMed:20935635, PubMed:21063390, PubMed:23355470, PubMed:23601106, PubMed:23602554, PubMed:25556658, PubMed:32351706, PubMed:26829474, PubMed:30704899, PubMed:34741373). CDK1/CDC2-cyclin-B controls pronuclear union in interphase fertilized eggs (PubMed:18480403, PubMed:20360007). Essential for early stages of embryonic development (PubMed:18480403, PubMed:20360007). During G2 and early mitosis, CDC25A/B/C-mediated dephosphorylation activates CDK1/cyclin complexes which phosphorylate several substrates that trigger at least centrosome separation, Golgi dynamics, nuclear envelope breakdown and chromosome condensation (PubMed:2344612, PubMed:2188730, PubMed:18480403, PubMed:20360007, PubMed:30139873). Once chromosomes are condensed and aligned at the metaphase plate, CDK1 activity is switched off by WEE1- and PKMYT1-mediated phosphorylation to allow sister chromatid separation, chromosome decondensation, reformation of the nuclear envelope and cytokinesis (PubMed:18480403, PubMed:20360007). Phosphorylates KRT5 during prometaphase and metaphase (By similarity). Inactivated by PKR/EIF2AK2- and WEE1-mediated phosphorylation upon DNA damage to stop cell cycle and genome replication at the G2 checkpoint thus facilitating DNA repair (PubMed:20360007). Reactivated after successful DNA repair through WIP1-dependent signaling leading to CDC25A/B/C-mediated dephosphorylation and restoring cell cycle progression (PubMed:20395957). Catalyzes lamin (LMNA, LMNB1 and LMNB2) phosphorylation at the onset of mitosis, promoting nuclear envelope breakdown (PubMed:2344612, PubMed:2188730, PubMed:37788673). In proliferating cells, CDK1-mediated FOXO1 phosphorylation at the G2-M phase represses FOXO1 interaction with 14-3-3 proteins and thereby promotes FOXO1 nuclear accumulation and transcription factor activity, leading to cell death of postmitotic neurons (PubMed:18356527). The phosphorylation of beta-tubulins regulates microtubule dynamics during mitosis (PubMed:16371510). NEDD1 phosphorylation promotes PLK1-mediated NEDD1 phosphorylation and subsequent targeting of the gamma-tubulin ring complex (gTuRC) to the centrosome, an important step for spindle formation (PubMed:19509060). In addition, CC2D1A phosphorylation regulates CC2D1A spindle pole localization and association with SCC1/RAD21 and centriole cohesion during mitosis (PubMed:20171170). The phosphorylation of Bcl-xL/BCL2L1 after prolongated G2 arrest upon DNA damage triggers apoptosis (PubMed:19917720). In contrast, CASP8 phosphorylation during mitosis prevents its activation by proteolysis and subsequent apoptosis (PubMed:20937773). This phosphorylation occurs in cancer cell lines, as well as in primary breast tissues and lymphocytes (PubMed:20937773). EZH2 phosphorylation promotes H3K27me3 maintenance and epigenetic gene silencing (PubMed:20935635). CALD1 phosphorylation promotes Schwann cell migration during peripheral nerve regeneration (By similarity). CDK1-cyclin-B complex phosphorylates NCKAP5L and mediates its dissociation from centrosomes during mitosis (PubMed:26549230). Regulates the amplitude of the cyclic expression of the core clock gene BMAL1 by phosphorylating its transcriptional repressor NR1D1, and this phosphorylation is necessary for SCF(FBXW7)- mediated ubiquitination and proteasomal degradation of NR1D1 (PubMed:27238018). Phosphorylates EML3 at 'Thr-881' which is essential for its interaction with HAUS augmin-like complex and TUBG1 (PubMed:30723163). Phosphorylates CGAS during mitosis, leading to its inhibition, thereby preventing CGAS activation by self DNA during mitosis (PubMed:32351706). |
| Cellular Location | Nucleus {ECO:0000250|UniProtKB:P11440}. Cytoplasm {ECO:0000250|UniProtKB:P11440}. Mitochondrion. Cytoplasm, cytoskeleton, microtubule organizing center, centrosome. Cytoplasm, cytoskeleton, spindle. Note=Cytoplasmic during the interphase Colocalizes with SIRT2 on centrosome during prophase and on splindle fibers during metaphase of the mitotic cell cycle. Reversibly translocated from cytoplasm to nucleus when phosphorylated before G2-M transition when associated with cyclin-B1. Accumulates in mitochondria in G2-arrested cells upon DNA-damage |
| Tissue Location | [Isoform 2]: Found in breast cancer tissues. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
References
1. Mol Biol Cell. 2008 Aug;19(8):3536-43. 2. Eur J Cancer. 2009 May;45(8):1466-73.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.