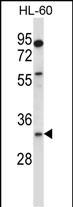
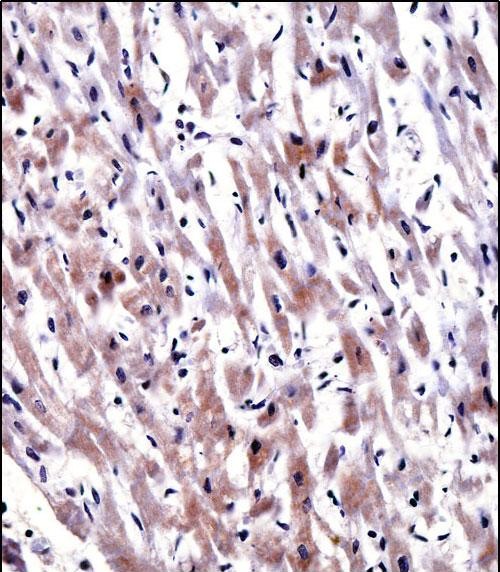
EGLN3 Antibody (C-term)
Affinity Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| IHC-P, WB, E |
|---|---|
| Primary Accession | Q9H6Z9 |
| Other Accession | Q62630, Q91UZ4, NP_071356.1 |
| Reactivity | Human |
| Predicted | Mouse, Rat |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 27261 Da |
| Antigen Region | 203-231 aa |
| Gene ID | 112399 |
|---|---|
| Other Names | Egl nine homolog 3, HPH-1, Hypoxia-inducible factor prolyl hydroxylase 3, HIF-PH3, HIF-prolyl hydroxylase 3, HPH-3, Prolyl hydroxylase domain-containing protein 3, PHD3, EGLN3 |
| Target/Specificity | This EGLN3 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 203-231 amino acids from the C-terminal region of human EGLN3. |
| Dilution | WB~~1:1000 IHC-P~~1:10~50 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | EGLN3 Antibody (C-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | EGLN3 {ECO:0000303|PubMed:16098468, ECO:0000312|HGNC:HGNC:14661} |
|---|---|
| Function | Prolyl hydroxylase that mediates hydroxylation of proline residues in target proteins, such as PKM, TELO2, ATF4 and HIF1A (PubMed:19584355, PubMed:21620138, PubMed:21483450, PubMed:22797300, PubMed:20978507, PubMed:21575608). Target proteins are preferentially recognized via a LXXLAP motif. Cellular oxygen sensor that catalyzes, under normoxic conditions, the post-translational formation of 4- hydroxyproline in hypoxia-inducible factor (HIF) alpha proteins (PubMed:11595184, PubMed:12181324). Hydroxylates a specific proline found in each of the oxygen-dependent degradation (ODD) domains (N- terminal, NODD, and C-terminal, CODD) of HIF1A (PubMed:11595184, PubMed:12181324). Also hydroxylates HIF2A (PubMed:11595184, PubMed:12181324). Has a preference for the CODD site for both HIF1A and HIF2A (PubMed:11595184, PubMed:12181324). Hydroxylation on the NODD site by EGLN3 appears to require prior hydroxylation on the CODD site (PubMed:11595184, PubMed:12181324). Hydroxylated HIFs are then targeted for proteasomal degradation via the von Hippel-Lindau ubiquitination complex (PubMed:11595184, PubMed:12181324). Under hypoxic conditions, the hydroxylation reaction is attenuated allowing HIFs to escape degradation resulting in their translocation to the nucleus, heterodimerization with HIF1B, and increased expression of hypoxy- inducible genes (PubMed:11595184, PubMed:12181324). ELGN3 is the most important isozyme in limiting physiological activation of HIFs (particularly HIF2A) in hypoxia. Also hydroxylates PKM in hypoxia, limiting glycolysis (PubMed:21620138, PubMed:21483450). Under normoxia, hydroxylates and regulates the stability of ADRB2 (PubMed:19584355). Regulator of cardiomyocyte and neuronal apoptosis. In cardiomyocytes, inhibits the anti-apoptotic effect of BCL2 by disrupting the BAX-BCL2 complex (PubMed:20849813). In neurons, has a NGF-induced proapoptotic effect, probably through regulating CASP3 activity (PubMed:16098468). Also essential for hypoxic regulation of neutrophilic inflammation (PubMed:21317538). Plays a crucial role in DNA damage response (DDR) by hydroxylating TELO2, promoting its interaction with ATR which is required for activation of the ATR/CHK1/p53 pathway (PubMed:22797300). Also mediates hydroxylation of ATF4, leading to decreased protein stability of ATF4 (Probable). |
| Cellular Location | Nucleus. Cytoplasm Note=Colocalizes with WDR83 in the cytoplasm {ECO:0000250|UniProtKB:Q62630} |
| Tissue Location | Widely expressed at low levels. Expressed at higher levels in adult heart (cardiac myocytes, aortic endothelial cells and coronary artery smooth muscle), lung and placenta, and in fetal spleen, heart and skeletal muscle. Also expressed in pancreas. Localized to pancreatic acini and islet cells. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
EGLN3 catalyzes the post-translational formation of 4-hydroxyproline in hypoxia-inducible factor (HIF) alpha proteins. Hydroxylates HIF-1 alpha at 'Pro-564', and HIF-2 alpha. Functions as a cellular oxygen sensor and, under normoxic conditions, targets HIF through the hydroxylation for proteasomal degradation via the von Hippel-Lindau ubiquitination complex. May play a role in cell growth regulation in muscle cells and in apoptosis in neuronal tissue. Promotes cell death through a caspase-dependent mechanism (By similarity).
References
Sato, M., et al. Exp. Cell Res. 316(17):2871-2882(2010)
Rose, J.E., et al. Mol. Med. 16 (7-8), 247-253 (2010) :
Xue, J., et al. Gastroenterology 138(2):606-615(2010)
Henze, A.T., et al. Cancer Res. 70(1):357-366(2010)
Hatzimichael, E., et al. Eur. J. Haematol. 84(1):47-51(2010)
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.














 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.