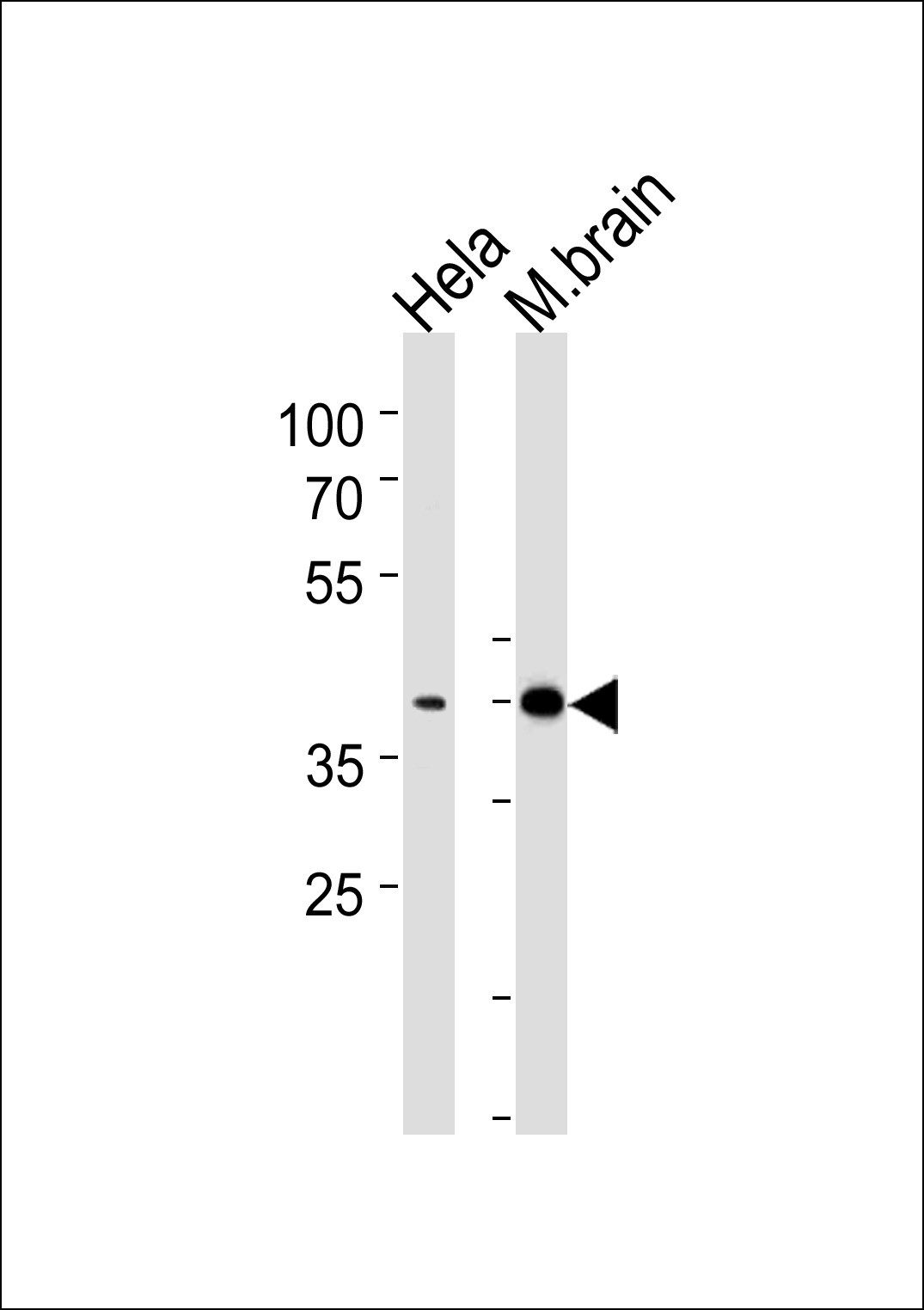
ATF2 Antibody (Center T339)
Affinity Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, E |
|---|---|
| Primary Accession | P15336 |
| Other Accession | NP_001871.2 |
| Reactivity | Human, Mouse |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 54537 Da |
| Antigen Region | 318-347 aa |
| Gene ID | 1386 |
|---|---|
| Other Names | Cyclic AMP-dependent transcription factor ATF-2, cAMP-dependent transcription factor ATF-2, Activating transcription factor 2, Cyclic AMP-responsive element-binding protein 2, CREB-2, cAMP-responsive element-binding protein 2, HB16, Histone acetyltransferase ATF2, cAMP response element-binding protein CRE-BP1, ATF2, CREB2, CREBP1 |
| Target/Specificity | This ATF2 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 318-347 amino acids from the Central region of human ATF2. |
| Dilution | WB~~1:1000 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | ATF2 Antibody (Center T339) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | ATF2 |
|---|---|
| Synonyms | CREB2, CREBP1 |
| Function | Transcriptional activator which regulates the transcription of various genes, including those involved in anti-apoptosis, cell growth, and DNA damage response. Dependent on its binding partner, binds to CRE (cAMP response element) consensus sequences (5'-TGACGTCA- 3') or to AP-1 (activator protein 1) consensus sequences (5'-TGACTCA- 3'). In the nucleus, contributes to global transcription and the DNA damage response, in addition to specific transcriptional activities that are related to cell development, proliferation and death. In the cytoplasm, interacts with and perturbs HK1- and VDAC1-containing complexes at the mitochondrial outer membrane, thereby impairing mitochondrial membrane potential, inducing mitochondrial leakage and promoting cell death. The phosphorylated form (mediated by ATM) plays a role in the DNA damage response and is involved in the ionizing radiation (IR)-induced S phase checkpoint control and in the recruitment of the MRN complex into the IR-induced foci (IRIF). Exhibits histone acetyltransferase (HAT) activity which specifically acetylates histones H2B and H4 in vitro (PubMed:10821277). In concert with CUL3 and RBX1, promotes the degradation of KAT5 thereby attenuating its ability to acetylate and activate ATM. Can elicit oncogenic or tumor suppressor activities depending on the tissue or cell type. |
| Cellular Location | Nucleus. Cytoplasm. Mitochondrion outer membrane. Note=Shuttles between the cytoplasm and the nucleus and heterodimerization with JUN is essential for the nuclear localization Localization to the cytoplasm is observed under conditions of cellular stress and in disease states. Localizes at the mitochondrial outer membrane in response to genotoxic stress. Phosphorylation at Thr-52 is required for its nuclear localization and negatively regulates its mitochondrial localization. Co-localizes with the MRN complex in the IR-induced foci (IRIF) |
| Tissue Location | Ubiquitously expressed, with more abundant expression in the brain |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
This gene encodes a transcription factor that is a member of the leucine zipper family of DNA binding proteins. This protein binds to the cAMP-responsive element (CRE), an octameric palindrome. The protein forms a homodimer or heterodimer with c-Jun and stimulates CRE-dependent transcription. The protein is also a histone acetyltransferase (HAT) that specifically acetylates histones H2B and H4 in vitro; thus it may represent a class of sequence-specific factors that activate transcription by direct effects on chromatin components. Additional transcript variants have been identified but their biological validity has not been determined.
References
Bailey, S.D., et al. Diabetes Care 33(10):2250-2253(2010)
Carrillo, R.J., et al. Biophys. Chem. 151(3):149-154(2010)
Lee, S.H., et al. Oncogene 29(37):5182-5192(2010)
Salameh, A., et al. J. Biol. Chem. 285(30):23096-23104(2010)
Bolat, I., et al. Folia Histochem. Cytobiol. 48(2):197-201(2010)
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.