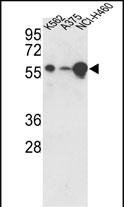
USP3 Antibody (C-term)
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| IHC-P, WB, E |
|---|---|
| Primary Accession | Q9Y6I4 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 58897 Da |
| Antigen Region | 489-519 aa |
| Gene ID | 9960 |
|---|---|
| Other Names | Ubiquitin carboxyl-terminal hydrolase 3, Deubiquitinating enzyme 3, Ubiquitin thioesterase 3, Ubiquitin-specific-processing protease 3, USP3 |
| Target/Specificity | This USP3 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 489-519 amino acids from the C-terminal region of human USP3. |
| Dilution | WB~~1:1000 IHC-P~~1:50~100 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis against PBS. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | USP3 Antibody (C-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | USP3 |
|---|---|
| Function | Hydrolase that deubiquitinates monoubiquitinated target proteins such as histone H2A and H2B. Required for proper progression through S phase and subsequent mitotic entry. May regulate the DNA damage response (DDR) checkpoint through deubiquitination of H2A at DNA damage sites. Associates with the chromatin. |
| Cellular Location | Nucleus. Note=Localizes preferentially with monoubiquitinated H2A to chromatin |
| Tissue Location | Expressed in all tissues examined, with strongest expression in pancreas |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Modification of target proteins by ubiquitin participates in a wide array of biological functions. Proteins destined for degradation or processing via the 26 S proteasome are coupled to multiple copies of ubiquitin. However, attachment of ubiquitin or ubiquitin-related molecules may also result in changes in subcellular distribution or modification of protein activity. An additional level of ubiquitin regulation, deubiquitination, is catalyzed by proteases called deubiquitinating enzymes, which fall into four distinct families. Ubiquitin C-terminal hydrolases, ubiquitin-specific processing proteases (USPs),1 OTU-domain ubiquitin-aldehyde-binding proteins, and Jab1/Pad1/MPN-domain-containing metallo-enzymes. Among these four families, USPs represent the most widespread and represented deubiquitinating enzymes across evolution. USPs tend to release ubiquitin from a conjugated protein. They display similar catalytic domains containing conserved Cys and His boxes but divergent N-terminal and occasionally C-terminal extensions, which are thought to function in substrate recognition, subcellular localization, and protein-protein interactions.
References
Puente, X.S., et al., Nat. Rev. Genet. 4(7):544-558 (2003). Sloper-Mould, K.E., et al., J. Biol. Chem. 274(38):26878-26884 (1999).
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.














 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.