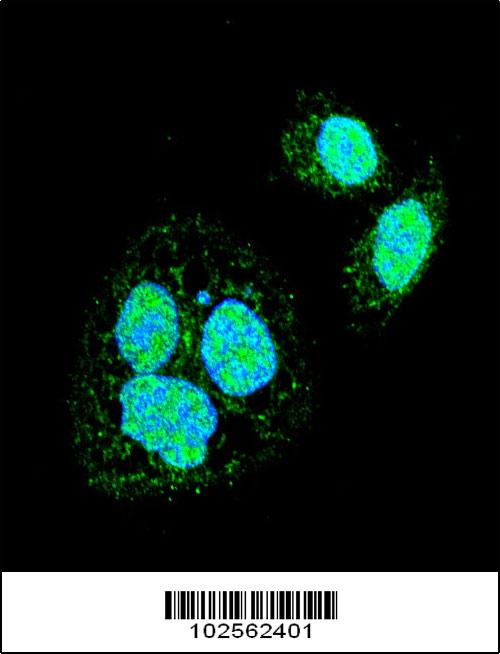
HSF1 Sumoylation Site Antibody
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IF, E |
|---|---|
| Primary Accession | Q00613 |
| Other Accession | Q08DJ8 |
| Reactivity | Human, Mouse |
| Predicted | Bovine |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 57260 Da |
| Antigen Region | 278-309 aa |
| Gene ID | 3297 |
|---|---|
| Other Names | Heat shock factor protein 1, HSF 1, Heat shock transcription factor 1, HSTF 1, HSF1, HSTF1 |
| Target/Specificity | This HSF1 Sumoylation Site antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 278-309 amino acids from human HSF1 Sumoylation Site. |
| Dilution | IF~~1:10~50 WB~~1:1000 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis against PBS. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | HSF1 Sumoylation Site Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | HSF1 (HGNC:5224) |
|---|---|
| Synonyms | HSTF1 |
| Function | Functions as a stress-inducible and DNA-binding transcription factor that plays a central role in the transcriptional activation of the heat shock response (HSR), leading to the expression of a large class of molecular chaperones, heat shock proteins (HSPs), that protect cells from cellular insult damage (PubMed:1871105, PubMed:11447121, PubMed:1986252, PubMed:7760831, PubMed:7623826, PubMed:8946918, PubMed:8940068, PubMed:9341107, PubMed:9121459, PubMed:9727490, PubMed:9499401, PubMed:9535852, PubMed:12659875, PubMed:12917326, PubMed:15016915, PubMed:25963659, PubMed:26754925, PubMed:18451878). In unstressed cells, is present in a HSP90-containing multichaperone complex that maintains it in a non-DNA-binding inactivated monomeric form (PubMed:9727490, PubMed:11583998, PubMed:16278218). Upon exposure to heat and other stress stimuli, undergoes homotrimerization and activates HSP gene transcription through binding to site-specific heat shock elements (HSEs) present in the promoter regions of HSP genes (PubMed:1871105, PubMed:1986252, PubMed:8455624, PubMed:7935471, PubMed:7623826, PubMed:8940068, PubMed:9727490, PubMed:9499401, PubMed:10359787, PubMed:11583998, PubMed:12659875, PubMed:16278218, PubMed:25963659, PubMed:26754925). Upon heat shock stress, forms a chromatin-associated complex with TTC5/STRAP and p300/EP300 to stimulate HSR transcription, therefore increasing cell survival (PubMed:18451878). Activation is reversible, and during the attenuation and recovery phase period of the HSR, returns to its unactivated form (PubMed:11583998, PubMed:16278218). Binds to inverted 5'-NGAAN-3' pentamer DNA sequences (PubMed:1986252, PubMed:26727489). Binds to chromatin at heat shock gene promoters (PubMed:25963659). Activates transcription of transcription factor FOXR1 which in turn activates transcription of the heat shock chaperones HSPA1A and HSPA6 and the antioxidant NADPH-dependent reductase DHRS2 (PubMed:34723967). Also serves several other functions independently of its transcriptional activity. Involved in the repression of Ras-induced transcriptional activation of the c-fos gene in heat-stressed cells (PubMed:9341107). Positively regulates pre-mRNA 3'-end processing and polyadenylation of HSP70 mRNA upon heat-stressed cells in a symplekin (SYMPK)-dependent manner (PubMed:14707147). Plays a role in nuclear export of stress- induced HSP70 mRNA (PubMed:17897941). Plays a role in the regulation of mitotic progression (PubMed:18794143). Also plays a role as a negative regulator of non-homologous end joining (NHEJ) repair activity in a DNA damage-dependent manner (PubMed:26359349). Involved in stress-induced cancer cell proliferation in a IER5-dependent manner (PubMed:26754925). |
| Cellular Location | Nucleus. Cytoplasm. Nucleus, nucleoplasm. Cytoplasm, perinuclear region. Cytoplasm, cytoskeleton, spindle pole. Cytoplasm, cytoskeleton, microtubule organizing center, centrosome Chromosome, centromere, kinetochore Note=The monomeric form is cytoplasmic in unstressed cells (PubMed:8455624, PubMed:26159920). Predominantly nuclear protein in both unstressed and heat shocked cells (PubMed:10413683, PubMed:10359787). Translocates in the nucleus upon heat shock (PubMed:8455624). Nucleocytoplasmic shuttling protein (PubMed:26159920). Colocalizes with IER5 in the nucleus (PubMed:27354066). Colocalizes with BAG3 to the nucleus upon heat stress (PubMed:8455624, PubMed:26159920). Localizes in subnuclear granules called nuclear stress bodies (nSBs) upon heat shock (PubMed:11447121, PubMed:11514557, PubMed:10359787, PubMed:25963659, PubMed:10747973, PubMed:24581496, PubMed:19229036). Colocalizes with SYMPK and SUMO1 in nSBs upon heat shock (PubMed:11447121, PubMed:12665592, PubMed:11514557, PubMed:14707147, PubMed:10359787) Colocalizes with PRKACA/PKA in the nucleus and nSBs upon heat shock (PubMed:21085490). Relocalizes from the nucleus to the cytoplasm during the attenuation and recovery phase period of the heat shock response (PubMed:26159920). Translocates in the cytoplasm in a YWHAE- and XPO1/CRM1-dependent manner (PubMed:12917326). Together with histone H2AX, redistributed in discrete nuclear DNA damage-induced foci after ionizing radiation (IR) (PubMed:26359349). Colocalizes with calcium- responsive transactivator SS18L1 at kinetochore region on the mitotic chromosomes (PubMed:18794143). Colocalizes with gamma tubulin at centrosome (PubMed:18794143). Localizes at spindle pole in metaphase (PubMed:18794143). Colocalizes with PLK1 at spindle poles during prometaphase (PubMed:18794143). |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Heat shock transcription factor 1 (HSF1) mediates the induction of heat shock protein gene expression in cells exposed to elevated temperature and other stress conditions. In response to stress, HSF1 acquires DNA-binding ability and localizes to nuclear stress granules. SUMO modification of HSF1 converts HSF1 to the DNA-binding form. HSF1 colocalizes with SUMO-1 in nuclear stress granules, which is prevented by mutation of the HSF1 lysine targeted for sumoylation.
References
Hilgarth, et al., Biochem Biophys Res Commun. 2003 Mar 28;303(1):196-200.
He, H., et al., J. Biol. Chem. 278(37):35465-35475 (2003).
Wang, X., et al., Mol. Cell. Biol. 23(17):6013-6026 (2003).
Ignatenko, N.A., et al., Exp. Cell Res. 288(1):1-8 (2003).
Soncin, F., et al., Biochem. Biophys. Res. Commun. 303(2):700-706 (2003).
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.