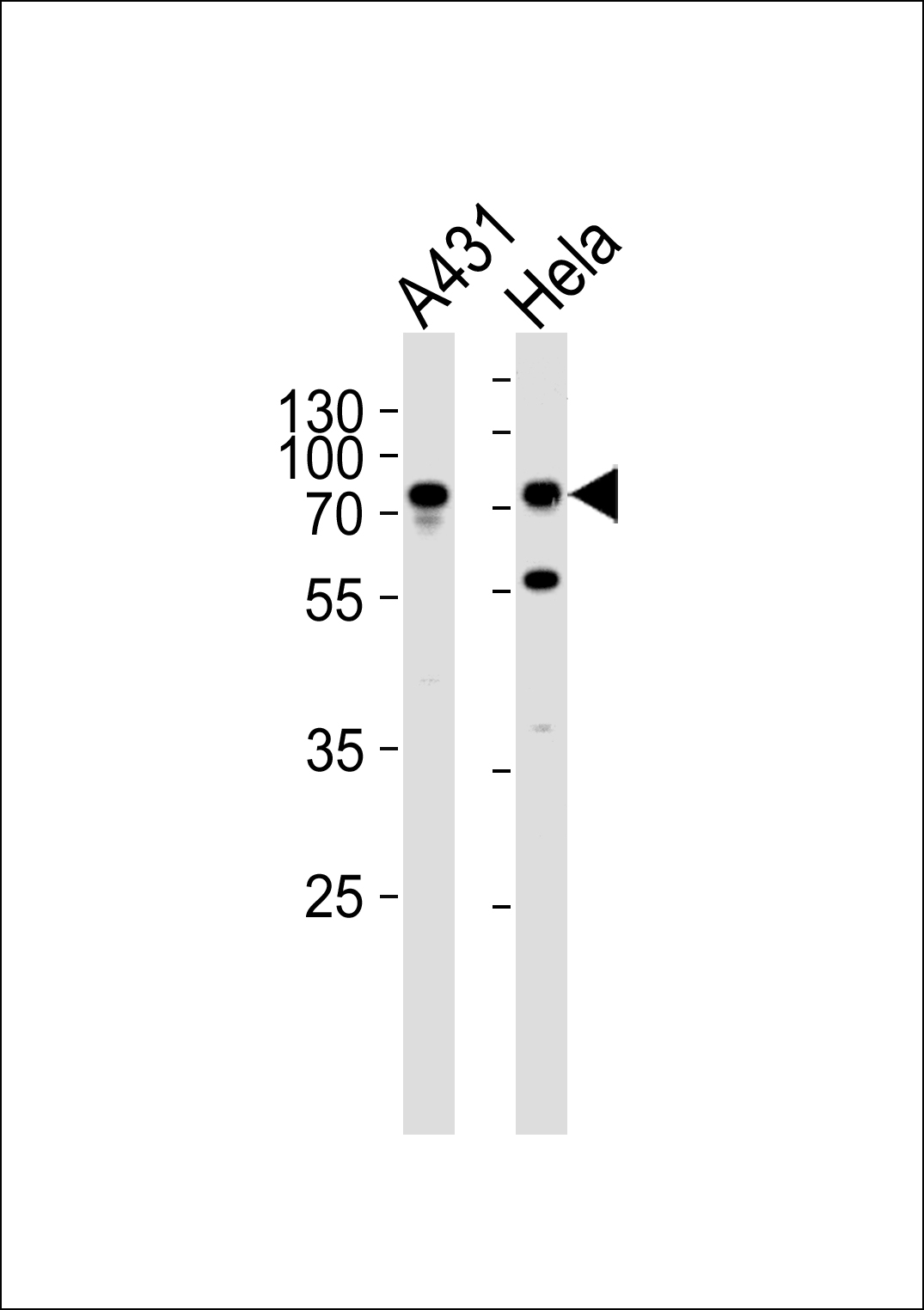
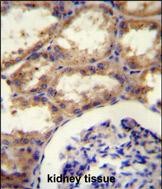
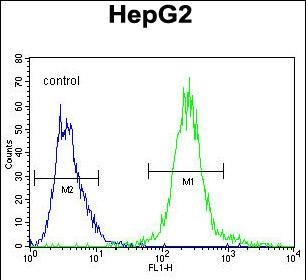
SEPT9 Antibody (C-term)
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS: 2
- PROTOCOLS
- BACKGROUND

Application
| WB, FC, IHC-P, E |
|---|---|
| Primary Accession | Q9UHD8 |
| Other Accession | NP_006631.2 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 65401 Da |
| Antigen Region | 557-586 aa |
| Gene ID | 10801 |
|---|---|
| Other Names | Septin-9, MLL septin-like fusion protein MSF-A, MLL septin-like fusion protein, Ovarian/Breast septin, Ov/Br septin, Septin D1, SEPT9, KIAA0991, MSF |
| Target/Specificity | This SEPT9 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 57-85 amino acids from the C-terminal region of human SEPT9. |
| Dilution | WB~~1:1000 IHC-P~~1:50~100 FC~~1:10~50 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | SEPT9 Antibody (C-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | SEPTIN9 (HGNC:7323) |
|---|---|
| Synonyms | KIAA0991, MSF, SEPT9 |
| Function | Filament-forming cytoskeletal GTPase (By similarity). May play a role in cytokinesis (Potential). May play a role in the internalization of 2 intracellular microbial pathogens, Listeria monocytogenes and Shigella flexneri. |
| Cellular Location | Cytoplasm, cytoskeleton. Note=In an epithelial cell line, concentrates at cell-cell contact areas. After TGF-beta1 treatment and induction of epithelial to mesenchymal transition, colocalizes partly with actin stress fibers. During bacterial infection, displays a collar shape structure next to actin at the pole of invading bacteria |
| Tissue Location | Widely expressed. Isoforms are differentially expressed in testes, kidney, liver heart, spleen, brain, peripheral blood leukocytes, skeletal muscle and kidney. Specific isoforms appear to demonstrate tissue specificity. Isoform 5 is the most highly expressed in fetal tissue. Isoform 1 is detected in all tissues except the brain and thymus, while isoform 2, isoform 3, and isoform 4 are detected at low levels in approximately half of the fetal tissues |

Provided below are standard protocols that you may find useful for product applications.
Background
The maf oncogene was identified by structural analysis of the AS42 avian transforming retrovirus genome. The Maf family is divided into two subclasses, large Mafs (vMaf, cMaf, MafB and Nrl) and small Mafs (MafF, MafK, and MafG). Both subclasses contain leucinezipper motifs, which allow homodimerization as well as heterodimerization with a variety of other bZip transcription factors. Large Mafs also contain an acidic transactivation domain absent in the small Maf proteins. Although they do not possess inherent transactivation activity, small Maf proteins can act as positive regulators of transcription by targeting transcriptionally active dimerization partners to specific DNA regulatory elements. Conversely, small Mafs can act also as negative regulators of transcription by recruiting transcriptional repressors or by forming homodimers that can replace active dimers. Human MafF was isolated in a yeast one-hybrid system from a human myometrium cDNA library. Human MAFF encodes a 164 amino acids proten. Like other small MAFF proteins, it contains an extended leucine zipper structure and lacks an N-terminal transactivating domain. The three small Maf proteins have been implicated in a number of physiological processes, including development, differentiation, haematopoiesis and stress response. Interestingly, these three proteins regulate the stress response via different mechanisms.
References
Proc. Natl. Acad. Sci. U.S.A. 96:6428-6433(1999). Cancer Res. 60: 4729-4734, 2000. Oncogene 20: 5930-5939, 2001.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.














 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.