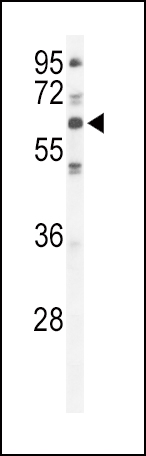
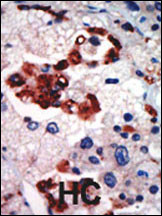
PKC iota Antibody (N-term)
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS: 3
- PROTOCOLS
- BACKGROUND

Application
| IHC-P, WB, E |
|---|---|
| Primary Accession | P41743 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 68262 Da |
| Antigen Region | 176-207 aa |
| Gene ID | 5584 |
|---|---|
| Other Names | Protein kinase C iota type, Atypical protein kinase C-lambda/iota, PRKC-lambda/iota, aPKC-lambda/iota, nPKC-iota, PRKCI, DXS1179E |
| Target/Specificity | This PKC iota antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 176-207 amino acids from the N-terminal region of human PKC iota. |
| Dilution | WB~~1:1000 IHC-P~~1:50~100 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis against PBS. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | PKC iota Antibody (N-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | PRKCI |
|---|---|
| Synonyms | DXS1179E |
| Function | Calcium- and diacylglycerol-independent serine/ threonine- protein kinase that plays a general protective role against apoptotic stimuli, is involved in NF-kappa-B activation, cell survival, differentiation and polarity, and contributes to the regulation of microtubule dynamics in the early secretory pathway. Is necessary for BCR-ABL oncogene-mediated resistance to apoptotic drug in leukemia cells, protecting leukemia cells against drug-induced apoptosis. In cultured neurons, prevents amyloid beta protein-induced apoptosis by interrupting cell death process at a very early step. In glioblastoma cells, may function downstream of phosphatidylinositol 3-kinase (PI(3)K) and PDPK1 in the promotion of cell survival by phosphorylating and inhibiting the pro-apoptotic factor BAD. Can form a protein complex in non-small cell lung cancer (NSCLC) cells with PARD6A and ECT2 and regulate ECT2 oncogenic activity by phosphorylation, which in turn promotes transformed growth and invasion. In response to nerve growth factor (NGF), acts downstream of SRC to phosphorylate and activate IRAK1, allowing the subsequent activation of NF-kappa-B and neuronal cell survival. Functions in the organization of the apical domain in epithelial cells by phosphorylating EZR. This step is crucial for activation and normal distribution of EZR at the early stages of intestinal epithelial cell differentiation. Forms a protein complex with LLGL1 and PARD6B independently of PARD3 to regulate epithelial cell polarity. Plays a role in microtubule dynamics in the early secretory pathway through interaction with RAB2A and GAPDH and recruitment to vesicular tubular clusters (VTCs). In human coronary artery endothelial cells (HCAEC), is activated by saturated fatty acids and mediates lipid-induced apoptosis. Involved in early synaptic long term potentiation phase in CA1 hippocampal cells and short term memory formation (By similarity). |
| Cellular Location | Cytoplasm. Membrane. Endosome Nucleus Note=Transported into the endosome through interaction with SQSTM1/p62 After phosphorylation by SRC, transported into the nucleus through interaction with KPNB1. Colocalizes with CDK7 in the cytoplasm and nucleus. Transported to vesicular tubular clusters (VTCs) through interaction with RAB2A. |
| Tissue Location | Predominantly expressed in lung and brain, but also expressed at lower levels in many tissues including pancreatic islets Highly expressed in non-small cell lung cancers |

Provided below are standard protocols that you may find useful for product applications.
Background
PKC iota belongs to the protein kinase C (PKC) family of serine/threonine protein kinases. The PKC family comprises at least eight members, which are differentially expressed and are involved in a wide variety of cellular processes. This protein kinase is calcium-independent and phospholipid-dependent. It is not activated by phorbolesters or diacylglycerol. This kinase can be recruited to vesicle tubular clusters (VTCs) by direct interaction with the small GTPase RAB2, where this kinase phosphorylates glyceraldehydes-3-phosphate dehydrogenase (GAPD/GAPDH) and plays a role in microtubule dynamics in the early secretory pathway. This kinase is found to be necessary for BCL-ABL-mediated resistance to drug-induced apoptosis and therefore protects leukemia cells against drug-induced apoptosis.
References
Zhang, J., et al., J. Biol. Chem. 279(21):22118-22123 (2004).
Roehrl, M.H., et al., J. Biomol. NMR 26(4):373-374 (2003).
Tisdale, E.J., J. Biol. Chem. 278(52):52524-52530 (2003).
Suzuki, A., et al., J. Biochem. 133(1):9-16 (2003).
Acevedo-Duncan, M., et al., Cell Prolif. 35(1):23-36 (2002).
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.














 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.