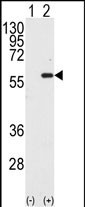
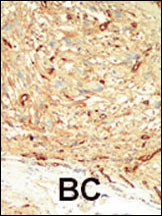
MST1 Antibody (C-term)
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS: 2
- PROTOCOLS
- BACKGROUND

Application
| IHC-P, WB, E |
|---|---|
| Primary Accession | Q13043 |
| Other Accession | Q5E9L6 |
| Reactivity | Human |
| Predicted | Bovine |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 55630 Da |
| Antigen Region | 385-415 aa |
| Gene ID | 6789 |
|---|---|
| Other Names | Serine/threonine-protein kinase 4, Mammalian STE20-like protein kinase 1, MST-1, STE20-like kinase MST1, Serine/threonine-protein kinase Krs-2, Serine/threonine-protein kinase 4 37kDa subunit, MST1/N, Serine/threonine-protein kinase 4 18kDa subunit, MST1/C, STK4, KRS2, MST1 |
| Target/Specificity | This MST1 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 385-415 amino acids from the C-terminal region of human MST1. |
| Dilution | WB~~1:1000 IHC-P~~1:50~100 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis against PBS. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | MST1 Antibody (C-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | STK4 |
|---|---|
| Synonyms | KRS2, MST1 |
| Function | Stress-activated, pro-apoptotic kinase which, following caspase-cleavage, enters the nucleus and induces chromatin condensation followed by internucleosomal DNA fragmentation. Key component of the Hippo signaling pathway which plays a pivotal role in organ size control and tumor suppression by restricting proliferation and promoting apoptosis. The core of this pathway is composed of a kinase cascade wherein STK3/MST2 and STK4/MST1, in complex with its regulatory protein SAV1, phosphorylates and activates LATS1/2 in complex with its regulatory protein MOB1, which in turn phosphorylates and inactivates YAP1 oncoprotein and WWTR1/TAZ. Phosphorylation of YAP1 by LATS2 inhibits its translocation into the nucleus to regulate cellular genes important for cell proliferation, cell death, and cell migration. STK3/MST2 and STK4/MST1 are required to repress proliferation of mature hepatocytes, to prevent activation of facultative adult liver stem cells (oval cells), and to inhibit tumor formation (By similarity). Phosphorylates 'Ser-14' of histone H2B (H2BS14ph) during apoptosis. Phosphorylates FOXO3 upon oxidative stress, which results in its nuclear translocation and cell death initiation. Phosphorylates MOBKL1A, MOBKL1B and RASSF2. Phosphorylates TNNI3 (cardiac Tn-I) and alters its binding affinity to TNNC1 (cardiac Tn-C) and TNNT2 (cardiac Tn-T). Phosphorylates FOXO1 on 'Ser-212' and regulates its activation and stimulates transcription of PMAIP1 in a FOXO1-dependent manner. Phosphorylates SIRT1 and inhibits SIRT1-mediated p53/TP53 deacetylation, thereby promoting p53/TP53 dependent transcription and apoptosis upon DNA damage. Acts as an inhibitor of PKB/AKT1. Phosphorylates AR on 'Ser-650' and suppresses its activity by intersecting with PKB/AKT1 signaling and antagonizing formation of AR- chromatin complexes. |
| Cellular Location | Cytoplasm. Nucleus. Note=The caspase-cleaved form cycles between the nucleus and cytoplasm |
| Tissue Location | Expressed in prostate cancer and levels increase from the normal to the malignant state (at protein level). Ubiquitously expressed. |

Provided below are standard protocols that you may find useful for product applications.
Background
MST1 is a cytoplasmic kinase that is structurally similar to the yeast Ste20p kinase, which acts upstream of the stress-induced mitogen-activated protein kinase cascade. The encoded protein can phosphorylate myelin basic protein and undergoes autophosphorylation. A caspase-cleaved fragment of the encoded protein has been shown to be capable of phosphorylating histone H2B. The particular phosphorylation catalyzed by this protein has been correlated with apoptosis, and it is possible that this protein induces the chromatin condensation observed in this process.
References
Cheung, W.L., et al., Cell 113(4):507-517 (2003).
Lin, Y., et al., J. Biol. Chem. 277(50):47991-48001 (2002).
De Souza, P.M., et al., Blood 99(9):3432-3438 (2002).
Ura, S., et al., Proc. Natl. Acad. Sci. U.S.A. 98(18):10148-10153 (2001).
Taylor, L.K., et al., Proc. Natl. Acad. Sci. U.S.A. 93(19):10099-10104 (1996).
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.















 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.