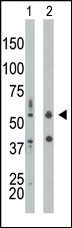
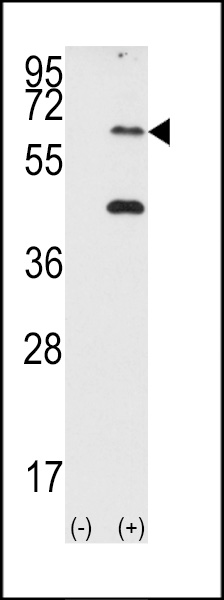
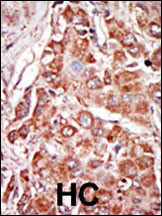
PKR (PRKR) Antibody (N-term)
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| IHC-P, WB, E |
|---|---|
| Primary Accession | P19525 |
| Other Accession | Q52M43 |
| Reactivity | Human, Mouse |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 62094 Da |
| Antigen Region | 11-42 aa |
| Gene ID | 5610 |
|---|---|
| Other Names | Interferon-induced, double-stranded RNA-activated protein kinase, Eukaryotic translation initiation factor 2-alpha kinase 2, eIF-2A protein kinase 2, Interferon-inducible RNA-dependent protein kinase, P1/eIF-2A protein kinase, Protein kinase RNA-activated, PKR, Protein kinase R, Tyrosine-protein kinase EIF2AK2, p68 kinase, EIF2AK2, PKR, PRKR |
| Target/Specificity | This PKR (PRKR) antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 11-42 amino acids from the N-terminal region of human PKR (PRKR). |
| Dilution | WB~~1:1000 IHC-P~~1:50~100 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis against PBS. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | PKR (PRKR) Antibody (N-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | EIF2AK2 |
|---|---|
| Synonyms | PKR, PRKR |
| Function | IFN-induced dsRNA-dependent serine/threonine-protein kinase that phosphorylates the alpha subunit of eukaryotic translation initiation factor 2 (EIF2S1/eIF-2-alpha) and plays a key role in the innate immune response to viral infection (PubMed:18835251, PubMed:19507191, PubMed:19189853, PubMed:21123651, PubMed:21072047, PubMed:22948139, PubMed:23229543, PubMed:22381929). Inhibits viral replication via the integrated stress response (ISR): EIF2S1/eIF-2- alpha phosphorylation in response to viral infection converts EIF2S1/eIF-2-alpha in a global protein synthesis inhibitor, resulting to a shutdown of cellular and viral protein synthesis, while concomitantly initiating the preferential translation of ISR-specific mRNAs, such as the transcriptional activator ATF4 (PubMed:19189853, PubMed:21123651, PubMed:22948139, PubMed:23229543). Exerts its antiviral activity on a wide range of DNA and RNA viruses including hepatitis C virus (HCV), hepatitis B virus (HBV), measles virus (MV) and herpes simplex virus 1 (HHV-1) (PubMed:11836380, PubMed:19189853, PubMed:20171114, PubMed:19840259, PubMed:21710204, PubMed:23115276, PubMed:23399035). Also involved in the regulation of signal transduction, apoptosis, cell proliferation and differentiation: phosphorylates other substrates including p53/TP53, PPP2R5A, DHX9, ILF3, IRS1 and the HHV-1 viral protein US11 (PubMed:11836380, PubMed:22214662, PubMed:19229320). In addition to serine/threonine- protein kinase activity, also has tyrosine-protein kinase activity and phosphorylates CDK1 at 'Tyr-4' upon DNA damage, facilitating its ubiquitination and proteasomal degradation (PubMed:20395957). Either as an adapter protein and/or via its kinase activity, can regulate various signaling pathways (p38 MAP kinase, NF-kappa-B and insulin signaling pathways) and transcription factors (JUN, STAT1, STAT3, IRF1, ATF3) involved in the expression of genes encoding pro-inflammatory cytokines and IFNs (PubMed:22948139, PubMed:23084476, PubMed:23372823). Activates the NF-kappa-B pathway via interaction with IKBKB and TRAF family of proteins and activates the p38 MAP kinase pathway via interaction with MAP2K6 (PubMed:10848580, PubMed:15121867, PubMed:15229216). Can act as both a positive and negative regulator of the insulin signaling pathway (ISP) (PubMed:20685959). Negatively regulates ISP by inducing the inhibitory phosphorylation of insulin receptor substrate 1 (IRS1) at 'Ser-312' and positively regulates ISP via phosphorylation of PPP2R5A which activates FOXO1, which in turn up-regulates the expression of insulin receptor substrate 2 (IRS2) (PubMed:20685959). Can regulate NLRP3 inflammasome assembly and the activation of NLRP3, NLRP1, AIM2 and NLRC4 inflammasomes (PubMed:22801494). Plays a role in the regulation of the cytoskeleton by binding to gelsolin (GSN), sequestering the protein in an inactive conformation away from actin (By similarity). |
| Cellular Location | Cytoplasm. Nucleus. Cytoplasm, perinuclear region. Note=Nuclear localization is elevated in acute leukemia, myelodysplastic syndrome (MDS), melanoma, breast, colon, prostate and lung cancer patient samples or cell lines as well as neurocytes from advanced Creutzfeldt- Jakob disease patients. |
| Tissue Location | Highly expressed in thymus, spleen and bone marrow compared to non-hematopoietic tissues such as small intestine, liver, or kidney tissues. Colocalizes with GSK3B and TAU in the Alzheimer disease (AD) brain. Elevated levels seen in breast and colon carcinomas, and which correlates with tumor progression and invasiveness or risk of progression. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Interferon-induced, double-stranded RNA-activated protein kinase (PRKR) is a serine-threonine kinase. Activation by dsRNAs leads to autophosphorylation of PRKR and allows the kinase to phosphorylate its natural substrate, the alpha subunit of eukaryotic protein synthesis initiation factor-2 (EIF2-alpha), leading to the inhibition of protein synthesis. Human gamma-interferon (IFNG) mRNA exploits localized activation of PRKR in the cell to regulate its own translation. IFNG mRNA activates PRKR through a pseudoknot in its 5-prime untranslated region. The HCV envelope protein E2 contains a sequence identical with phosphorylation sites of the interferon-inducible protein kinase PRKR and the translation initiation factor EIF2-alpha, a target of PRKR. E2 inhibits the kinase activity of PRKR and blocks its inhibitory effect on protein synthesis and cell growth, which provides one mechanism by which HCV may circumvent the antiviral effect of interferon. PRKR, which is involved in TLR signaling and mediates apoptosis in fibroblasts in response to viral infection and inflammatory cytokines, also activates IKK and NFKB, thereby suppressing apoptosis. Apoptosis induced by live pathogenic gram-positive and gram-negative bacteria requirs both TLR4 and PRKR, possibly representing a major mechanism for pathogenic bacteria that use specific virulence factors to avoid detection and destruction by the innate immune system. Roles for PRKR activation in Huntington disease and Fanconi anemia have also been suggested.
References
Peel, A.L., et al., Neurobiol. Dis. 14(1):52-62 (2003).
Pang, Q., et al., J. Biol. Chem. 277(51):49638-49643 (2002).
Kim, S.H., et al., Oncogene 21(57):8741-8748 (2002).
Takizawa, T., et al., Eur. J. Biochem. 269(24):6126-6132 (2002).
Peters, G.A., et al., J. Virol. 76(21):11054-11064 (2002).
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.