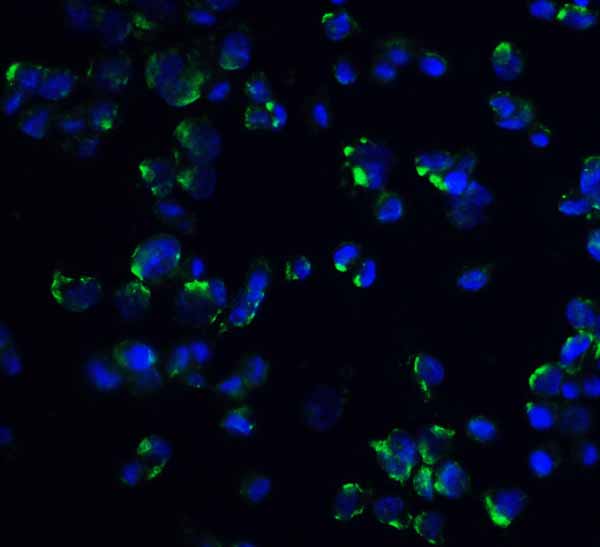
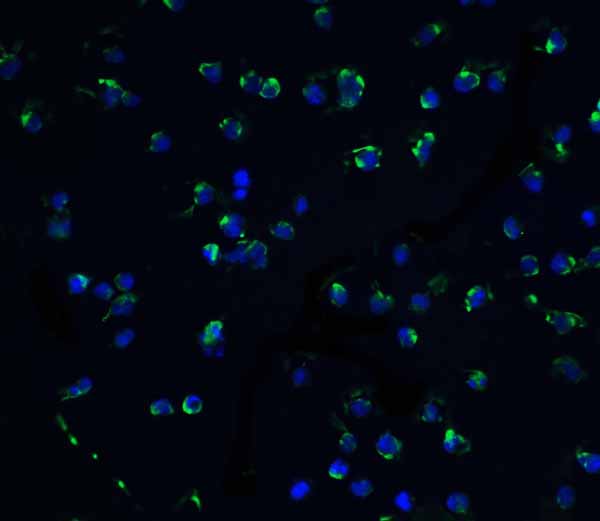
IL-1RAcP Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IF, ICC, E |
|---|---|
| Primary Accession | Q9NPH3 |
| Other Accession | NP_001161401, 3556 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
| Calculated MW | Predicted: 63 kDa Observed: 66 kDa |
| Application Notes | IL-1RAcP antibody can be used for detection of IL-1RAcP by Western blot at 1 μg/mL. Antibody can also be used for Immunohistochemistry starting at 2 μg/mL. For immunofluorescence start at 5 μg/mL. |
| Gene ID | 3556 |
|---|---|
| Other Names | IL-1RAcP Antibody: IL1R3, C3orf13, IL-1RAcP, IL1R3, Interleukin-1 receptor accessory protein, Interleukin-1 receptor 3, IL-1 receptor accessory protein, interleukin 1 receptor accessory protein |
| Target/Specificity | IL-1RAcP antibody was raised against a 16 amino acid peptide near the carboxy terminus of human IL-1RAcP. The immunogen is located within the last 50 amino acids of IL-1RAcP. |
| Reconstitution & Storage | IL-1RAcP antibody can be stored at 4℃ for three months and -20℃, stable for up to one year. As with all antibodies care should be taken to avoid repeated freeze thaw cycles. Antibodies should not be exposed to prolonged high temperatures. |
| Precautions | IL-1RAcP Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | IL1RAP |
|---|---|
| Synonyms | C3orf13, IL1R3 |
| Function | Coreceptor for IL1RL2 in the IL-36 signaling system (By similarity). Coreceptor with IL1R1 in the IL-1 signaling system. Associates with IL1R1 bound to IL1B to form the high affinity interleukin-1 receptor complex which mediates interleukin-1-dependent activation of NF-kappa-B and other pathways. Signaling involves the recruitment of adapter molecules such as TOLLIP, MYD88, and IRAK1 or IRAK2 via the respective TIR domains of the receptor/coreceptor subunits. Recruits TOLLIP to the signaling complex. Does not bind to interleukin-1 alone; binding of IL1RN to IL1R1, prevents its association with IL1R1 to form a signaling complex. The cellular response is modulated through a non-signaling association with the membrane IL1R2 decoy receptor. Coreceptor for IL1RL1 in the IL-33 signaling system. Can bidirectionally induce pre- and postsynaptic differentiation of neurons by trans-synaptically binding to PTPRD (By similarity). May play a role in IL1B-mediated costimulation of IFNG production from T-helper 1 (Th1) cells (Probable). |
| Cellular Location | [Isoform 1]: Cell membrane; Single-pass type I membrane protein [Isoform 3]: Secreted. |
| Tissue Location | Detected in liver, skin, placenta, thymus and lung. Isoform 4 is predominantly expressed in brain. Overexpressed on candidate chronic myeloid leukemia (CML) stem cells, hematopoietic stem cells and mononuclear cells of patients with acute myeloid leukemia (AML). Overexpressed in patients with chronic obstructive pulmonary disease (COPD). Expressed in T-helper 1 (Th1) and T-helper 2 (Th2) cell subsets (PubMed:10653850). |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
IL-1RAcP Antibody: The pro-inflammatory cytokine IL-1 induced cellular response requires two subunits of its receptor, IL-1 receptor I (IL-1RI) and IL-1 receptor accessory protein (IL-1RAcP). IL-1RAcP forms a complex with IL-1RI in response to IL-1 treatment. The IL-1 receptor-associated kinase (IRAK), which mediates activation of NF-κB inducing kinae (NIK) and of NF-κB, recruits to the IL-1R complex through IL-1RAcP. IL-1 activation of stress-activated protein kinase and of acid sphingomyelinase also requires IL-1RAcP. Like IL-1RI, IL-1RAcP subunit is essential for IL-1 mediated cellular response. IL-1RAcP is expressed in many tissues.
References
Greenfeder SA, Nunes P, Kwee L, et al. Molecular cloning and characterization of a second subunit of the interleukin 1 receptor complex. J. Biol. Chem. 1995; 270:13757-65.
Huang J, Gao X, Li S, et al. Recruitment of IRAK to the interleukin 1 receptor complex requires interleukin 1 receptor accessory protein. Proc. Natl. Acad. Sci. USA 1997; 94:12829-32.
Wesche H, Korherr C, Kracht M, et al. The interleukin-1 receptor accessory protein (IL-1RAcP) is essential for IL-1-induced activation of interleukin-1 receptor-associated kinase (IRAK) and stress-activated protein kinases (SAP kinases). J. Biol. Chem. 1997; 272:7727-31.
Hofmeister R, Wiegmann K, Korherr C, et al. Activation of acid sphingomyelinase by interleukin-1 (IL-1) requires the IL-1 receptor accessory protein. J. Biol. Chem. 1997; 272:27730-6.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.