PHAP III Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC-P, IF, E |
|---|---|
| Primary Accession | Q9BTT0 |
| Other Accession | NP_112182, 13569879 |
| Reactivity | Human, Mouse, Rat |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
| Calculated MW | 35 kDa |
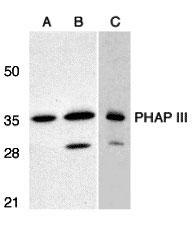
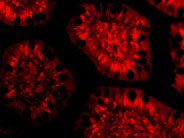
| Application Notes | PHAP III antibody can be used for detection of PHAP III by Western blot at 1 µg/mL. A band at approximately 35 kDa can be detected. Antibody can also be used for immunohistochemistry starting at 2 µg/mL. For immunofluorescence start at 10 µg/mL. |
| Gene ID | 81611 |
|---|---|
| Other Names | PHAP III Antibody: LANPL, LANP-L, Acidic leucine-rich nuclear phosphoprotein 32 family member E, LANP-like protein, acidic (leucine-rich) nuclear phosphoprotein 32 family, member E |
| Target/Specificity | ANP32E; PHAP III has no cross-reaction to PHAP I and PHAP I2a. |
| Reconstitution & Storage | PHAP III antibody can be stored at 4℃ for three months and -20℃, stable for up to one year. As with all antibodies care should be taken to avoid repeated freeze thaw cycles. Antibodies should not be exposed to prolonged high temperatures. |
| Precautions | PHAP III Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | ANP32E |
|---|---|
| Function | Histone chaperone that specifically mediates the genome-wide removal of histone H2A.Z/H2AZ1 from the nucleosome: removes H2A.Z/H2AZ1 from its normal sites of deposition, especially from enhancer and insulator regions. Not involved in deposition of H2A.Z/H2AZ1 in the nucleosome. May stabilize the evicted H2A.Z/H2AZ1-H2B dimer, thus shifting the equilibrium towards dissociation and the off-chromatin state (PubMed:24463511). Inhibits activity of protein phosphatase 2A (PP2A). Does not inhibit protein phosphatase 1. May play a role in cerebellar development and synaptogenesis. |
| Cellular Location | Cytoplasm. Nucleus. |
| Tissue Location | Expressed in peripheral blood leukocytes, colon, small intestine, prostate, thymus, spleen, skeletal muscle, liver and kidney. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
PHAP III Antibody: Apoptosis is related to many diseases and development. Caspase-9 plays a central role in cell death induced by a variety of apoptosis activators. Cytochrome c, after released from mitochondria, binds to Apaf-1, which forms an apoptosome that in turn binds to and activate procaspase-9. Activated caspase-9 cleaves and activates the effector caspases (caspase-3, -6 and -7), which are responsible for the proteolytic cleavage of many key proteins in apoptosis. The tumor suppressor putative HLA-DR-associated proteins (PHAPs) were recently identified as important regulators of mitochondrion apoptosis. PHAP appears to facilitate apoptosome-medicated caspase-9 activation and to stimulate the mitochondrial apoptotic pathway. PHAP was also shown to oppose both Ras- and Myc-medicated cell transformation.
References
Jiang X, Kim HE, Shu H, Zhao Y, Zhang H, Kofron J, Donnelly J, Burns D, Ng SC , Rosenberg S, Wang X. Distinctive roles of PHAP proteins and prothymosin-α in a death regulatory pathway. Science. 2003;299(5604):223-6.
Nicholson DW, Thornberry NA. Apoptosis. Life and death decisions. Science. 2003 10;299(5604):214-5.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.