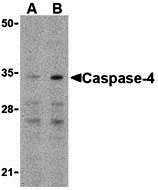
Caspase-4 Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC-P, IF, E |
|---|---|
| Primary Accession | P49662 |
| Other Accession | AAA86890, 886050 |
| Reactivity | Human, Mouse |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
| Calculated MW | 43262 Da |
| Application Notes | Casp-4 antibody can be used for the detection of Caspase-4 by Western blot at 0.5 - 1 µg/mL. Antibody can also be used for immunohistochemistry starting at 2 µg/mL. For immunofluorescence start at 10 µg/mL. |
| Gene ID | 837 |
|---|---|
| Other Names | Caspase-4 Antibody: TX, ICH-2, Mih1/TX, ICEREL-II, ICE(rel)II, ICH2, Caspase-4, Protease ICH-2, CASP-4, caspase 4, apoptosis-related cysteine peptidase |
| Target/Specificity | CASP4; Depending on cell lines or tissues used, other cleavage products may be observed. |
| Reconstitution & Storage | Caspase-4 antibody can be stored at 4℃ for three months and -20℃, stable for up to one year. As with all antibodies care should be taken to avoid repeated freeze thaw cycles. Antibodies should not be exposed to prolonged high temperatures. |
| Precautions | Caspase-4 Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | CASP4 {ECO:0000303|PubMed:15123740, ECO:0000312|HGNC:HGNC:1505} |
|---|---|
| Function | Inflammatory caspase that acts as the effector of the non- canonical inflammasome by mediating lipopolysaccharide (LPS)-induced pyroptosis (PubMed:25119034, PubMed:26375003, PubMed:34671164, PubMed:32109412, PubMed:37001519, PubMed:37993712, PubMed:37993714). Also indirectly activates the NLRP3 and NLRP6 inflammasomes (PubMed:7797510, PubMed:23516580, PubMed:26375003, PubMed:32109412). Acts as a thiol protease that cleaves a tetrapeptide after an Asp residue at position P1: catalyzes cleavage of CGAS, GSDMD and IL18 (PubMed:7797510, PubMed:15326478, PubMed:23516580, PubMed:26375003, PubMed:28314590, PubMed:32109412, PubMed:37993712, PubMed:37993714). Effector of the non-canonical inflammasome independently of NLRP3 inflammasome and CASP1: the non-canonical inflammasome promotes pyroptosis through GSDMD cleavage without involving secretion of cytokine IL1B (PubMed:25121752, PubMed:25119034, PubMed:26375003, PubMed:31268602, PubMed:32109412, PubMed:37993712, PubMed:37993714). In the non-canonical inflammasome, CASP4 is activated by direct binding to the lipid A moiety of LPS without the need of an upstream sensor (PubMed:25121752, PubMed:25119034, PubMed:29520027, PubMed:32510692, PubMed:32581219, PubMed:37993712). LPS-binding promotes CASP4 activation and CASP4-mediated cleavage of GSDMD and IL18, followed by IL18 secretion through the GSDMD pore, pyroptosis of infected cells and their extrusion into the gut lumen (PubMed:25121752, PubMed:25119034, PubMed:37993712, PubMed:37993714). Also indirectly promotes secretion of mature cytokines (IL1A and HMGB1) downstream of GSDMD-mediated pyroptosis via activation of the NLRP3 and NLRP6 inflammasomes (PubMed:26375003, PubMed:32109412). Involved in NLRP3-dependent CASP1 activation and IL1B secretion in response to non-canonical activators, such as UVB radiation or cholera enterotoxin (PubMed:22246630, PubMed:23516580, PubMed:24879791, PubMed:25964352, PubMed:26173988, PubMed:26174085, PubMed:26508369). Involved in NLRP6 inflammasome- dependent activation in response to lipoteichoic acid (LTA), a cell- wall component of Gram-positive bacteria, which leads to CASP1 activation and IL1B secretion (PubMed:33377178). Involved in LPS- induced IL6 secretion; this activity may not require caspase enzymatic activity (PubMed:26508369). The non-canonical inflammasome is required for innate immunity to cytosolic, but not vacuolar, bacteria (By similarity). Plays a crucial role in the restriction of S.typhimurium replication in colonic epithelial cells during infection (PubMed:25121752, PubMed:25964352). Pyroptosis limits bacterial replication, while cytokine secretion promotes the recruitment and activation of immune cells and triggers mucosal inflammation (PubMed:25121752, PubMed:26375003, PubMed:25964352). May also act as an activator of adaptive immunity in dendritic cells, following activation by oxidized phospholipid 1-palmitoyl-2-arachidonoyl- sn-glycero-3- phosphorylcholine, an oxidized phospholipid (oxPAPC) (By similarity). Involved in cell death induced by endoplasmic reticulum stress and by treatment with cytotoxic APP peptides found in Alzheimer's patient brains (PubMed:15123740, PubMed:22246630, PubMed:23661706). Cleavage of GSDMD is not strictly dependent on the consensus cleavage site but depends on an exosite interface on CASP4 that recognizes and binds the Gasdermin-D, C-terminal (GSDMD-CT) part (PubMed:32109412). Catalyzes cleavage and maturation of IL18; IL18 processing also depends of the exosite interface on CASP4 (PubMed:15326478, PubMed:37993712, PubMed:37993714). In contrast, it does not directly process IL1B (PubMed:7743998, PubMed:7797592, PubMed:7797510). During non-canonical inflammasome activation, cuts CGAS and may play a role in the regulation of antiviral innate immune activation (PubMed:28314590). |
| Cellular Location | Cytoplasm, cytosol. Endoplasmic reticulum membrane; Peripheral membrane protein; Cytoplasmic side. Mitochondrion Inflammasome. Secreted Note=Predominantly localizes to the endoplasmic reticulum (ER) Association with the ER membrane requires TMEM214 (PubMed:15123740) Released in the extracellular milieu by keratinocytes following UVB irradiation (PubMed:22246630). |
| Tissue Location | Widely expressed, including in keratinocytes and colonic and small intestinal epithelial cells (at protein level). Not detected in brain. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Caspase-4 Antibody: Caspases are a family of cysteine proteases that can be divided into the apoptotic and inflammatory caspase subfamilies. Unlike the apoptotic caspases, members of the inflammatory subfamily are generally not involved in cell death but are associated with the immune response to microbial pathogens. Members of this subfamily include caspase-1, -4, -5, and -12. Activation of these caspases results in the cleavage and activation of proinflammatory cytokines such as IL-1β and IL-18. Caspase-4 was initially identified as a homologous protein to Caspase-1 and the C. elegans Ced-3 which could induce apoptosis in transfected cells. More recent studies have shown that it can be activated by ER stress and has been suggested to be involved in multiple neuronal pathologies such as Alzheimer's disease.
References
Martinon F and Tschopp J. Inflammatory caspases: linking an intracellular innate immune system to autoinflammatory diseases. Cell 2004; 117:561-74.
Kuida K, Lippke JA, Ku G, et al. Altered cytokine export and apoptosis in mice deficient in interleukin-1 β converting enzyme. Science 1995; 267:2000-3.
Gracie JA, Robertson SE, and McInnes IB. Interleukin-18. J. Leukoc. Biol. 2003; 73:213-224.
Kamens J, Paskind M, Hugunin M, et al. Identification and characterization of ICH-2, a novel member of the interleukin-1 β-converting enzyme family of cysteine proteases. J. Biol. Chem. 1995; 270:15250-6.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.