PTEN Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, E, IP |
|---|---|
| Primary Accession | P60484 |
| Other Accession | P60484, 42560209 |
| Reactivity | Human, Mouse |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
| Calculated MW | 47166 Da |
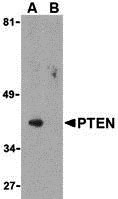
| Application Notes | PTEN antibody can be used for the detection of PTEN by Western blot at 1 µg/mL. |
| Gene ID | 5728 |
|---|---|
| Other Names | PTEN Antibody: BZS, DEC, CWS1, GLM2, MHAM, TEP1, MMAC1, PTEN1, 10q23del, Phosphatidylinositol 3, 4, 5-trisphosphate 3-phosphatase and dual-specificity protein phosphatase PTEN, Mutated in multiple advanced cancers 1, phosphatase and tensin homolog |
| Target/Specificity | PTEN; |
| Reconstitution & Storage | PTEN antibody can be stored at 4℃ for three months and -20℃, stable for up to one year. As with all antibodies care should be taken to avoid repeated freeze thaw cycles. Antibodies should not be exposed to prolonged high temperatures. |
| Precautions | PTEN Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | PTEN |
|---|---|
| Synonyms | MMAC1, TEP1 |
| Function | Dual-specificity protein phosphatase, dephosphorylating tyrosine-, serine- and threonine-phosphorylated proteins (PubMed:9187108, PubMed:9256433, PubMed:9616126). Also functions as a lipid phosphatase, removing the phosphate in the D3 position of the inositol ring of PtdIns(3,4,5)P3/phosphatidylinositol 3,4,5- trisphosphate, PtdIns(3,4)P2/phosphatidylinositol 3,4-diphosphate and PtdIns3P/phosphatidylinositol 3-phosphate with a preference for PtdIns(3,4,5)P3 (PubMed:9811831, PubMed:16824732, PubMed:26504226, PubMed:9593664). Furthermore, this enzyme can also act as a cytosolic inositol 3-phosphatase acting on Ins(1,3,4,5,6)P5/inositol 1,3,4,5,6 pentakisphosphate and possibly Ins(1,3,4,5)P4/1D-myo-inositol 1,3,4,5- tetrakisphosphate (PubMed:11418101, PubMed:15979280). Antagonizes the PI3K-AKT/PKB signaling pathway by dephosphorylating phosphoinositides and thereby modulating cell cycle progression and cell survival (PubMed:31492966, PubMed:37279284). The unphosphorylated form cooperates with MAGI2 to suppress AKT1 activation (PubMed:11707428). In motile cells, suppresses the formation of lateral pseudopods and thereby promotes cell polarization and directed movement (PubMed:22279049). Dephosphorylates tyrosine-phosphorylated focal adhesion kinase and inhibits cell migration and integrin-mediated cell spreading and focal adhesion formation (PubMed:22279049). Required for growth factor-induced epithelial cell migration; growth factor stimulation induces PTEN phosphorylation which changes its binding preference from the p85 regulatory subunit of the PI3K kinase complex to DLC1 and results in translocation of the PTEN-DLC1 complex to the posterior of migrating cells to promote RHOA activation (PubMed:26166433). Meanwhile, TNS3 switches binding preference from DLC1 to p85 and the TNS3-p85 complex translocates to the leading edge of migrating cells to activate RAC1 activation (PubMed:26166433). Plays a role as a key modulator of the AKT-mTOR signaling pathway controlling the tempo of the process of newborn neurons integration during adult neurogenesis, including correct neuron positioning, dendritic development and synapse formation (By similarity). Involved in the regulation of synaptic function in excitatory hippocampal synapses. Recruited to the postsynaptic membrane upon NMDA receptor activation, is required for the modulation of synaptic activity during plasticity. Enhancement of lipid phosphatase activity is able to drive depression of AMPA receptor-mediated synaptic responses, activity required for NMDA receptor-dependent long-term depression (LTD) (By similarity). May be a negative regulator of insulin signaling and glucose metabolism in adipose tissue. The nuclear monoubiquitinated form possesses greater apoptotic potential, whereas the cytoplasmic nonubiquitinated form induces less tumor suppressive ability (PubMed:10468583, PubMed:18716620). |
| Cellular Location | Cytoplasm. Nucleus. Nucleus, PML body. Cell projection, dendritic spine {ECO:0000250|UniProtKB:O54857}. Postsynaptic density {ECO:0000250|UniProtKB:O54857}. Note=Monoubiquitinated form is nuclear Nonubiquitinated form is cytoplasmic. Colocalized with PML and USP7 in PML nuclear bodies (PubMed:18716620). XIAP/BIRC4 promotes its nuclear localization (PubMed:19473982). Associares with the postsynaptic density in response to NMDAR activation (By similarity) {ECO:0000250|UniProtKB:O54857, ECO:0000269|PubMed:18716620, ECO:0000269|PubMed:19473982} |
| Tissue Location | Expressed at a relatively high level in all adult tissues, including heart, brain, placenta, lung, liver, muscle, kidney and pancreas. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
PTEN Antibody: PTEN (phosphatase and tensin homologue deleted on chromosome ten) is a dual-specificity phosphatase (with both protein and lipid phosphatase activity) first identified as a tumor suppressor gene. PTEN indirectly activates the AKT/PI3K pathway, an important signaling pathway for cell growth and proliferation by keeping levels of the second messenger PIP3 low, thereby preventing phosphoinositide-dependent kinase-1 (PDK-1) from phosphorylating and activating AKT. Expression of wild-type PTEN causes growth arrest in many cancer cell lines, but expression of a PTEN protein containing a mutation that blocks its lipid phosphatase activity failed to suppress growth of glioma cell lines suggesting that the tumor suppressive effect of PTEN is mediated solely by its lipid phosphatase activity. Other activities include the inhibition of insulin stimulated MAPK activation by blocking the insulin-receptor substrate (IRS)-1 phosphorylation and assembly of the IRS-1/Grb2/Sos complex.
References
Li J, Yen C, Liaw D, et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science1997; 275:1943-7.
Steck PA, Pershouse MA, Jasser SA, et al. Identification of a candidate tumor suppressor gene, MMAC1, a chromosome 10q23.3 that is mutated in multiple advanced cancers. Nat. Genet.1997; 15:356-62.
Wu X, Senechal K, Neshat MS, et al. The PTEN/MMAC1 tumor suppressor phosphatase functions as a negative regulator of the phosphoinositide 3-kinase/Akt pathway. Proc. Natl. Acad. Sci. USA1998; 95:15587-91.
Myers MP, Pass I, Batty IH, et al. The lipid phosphatase activity of PTEN is critical for its tumor suppressor function. Proc. Natl. Acad. Sci. USA1998; 95:13513-8.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.