H1N1 Neuraminidase Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| E |
|---|---|
| Primary Accession | B1AGS8, C4AL28 |
| Other Accession | ACA33620, ACQ76308, 168827347, 229535824 |
| Reactivity | Virus |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
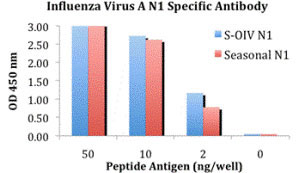
| Application Notes | Neuraminidase antibody can be used for the detection of the Neuraminidase protein from the seasonal influenza A (H1N1) and the novel swine influenza in ELISA. It will detect 2 ng of free peptide at 1 µg/mL. |
| Target/Specificity | NA; This antibody will recognize the Neuraminidase from the both the seasonal influenza (H1N1) and swine-origin influenza virus (A/California/14/2009 (H1N1)). |
|---|---|
| Reconstitution & Storage | H1N1 Neuraminidase antibody can be stored at 4℃ for three months and -20℃, stable for up to one year. As with all antibodies care should be taken to avoid repeated freeze thaw cycles. Antibodies should not be exposed to prolonged high temperatures. |
| Precautions | H1N1 Neuraminidase Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
H1N1 Neuraminidase Antibody: Influenza A virus is a major public health threat, killing more than 30, 000 people per year in the USA. In early 2009, a novel swine-origin influenza A (H1N1) virus was identified in specimens obtained from patients in Mexico and the United States. The virus spread quickly around the world and on June 11, 2009, the World Health Organization declared it a pandemic. Influenza A virus has one of sixteen possible Hemagglutinin (HA) surface proteins and one of nine possible Neuraminidase (NA) surface proteins. The Hemagglutinin protein facilitates viral attachment while Neuraminidase is involved in viral release. These proteins also elicit immune responses that prevent infection or independently reduce viral replication. The genetic make-up of this swine flu virus is unlike any other: it is an H1N1 strain that combines a triple assortment first identified in 1998 including human, swine, and avian influenza with two new pig H3N2 virus genes from Eurasia, themselves of recent human origin. The distinct antigenic properties of the new swine influenza virus compared with seasonal influenza A (H1N1) virus suggest that human immunity against new swine influenza virus is limited, although the age distribution of reported cases suggests some degree of protection in older age groups.
References
Thompson WW, Shay DK, Weintraub, et al. Mortality associated with influenza and respiratory syncytial virus in the United States. JAMA2003; 289:179-186.
Novel Swine-Origin Influenza A (H1N1) Virus Investigation Team, Dawood FS, Jain S, et al. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N. Engl. J. Med.2009; 360:2605-15.
Butler D. Swine flu goes global. Nature2009; 458:1082-3.
Morens DM, Taubenberger JK, and Fauci AS. The Persistent Legacy of the 1918 Influenza Virus. N. Engl. J. Med.2009; Jun 29.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.