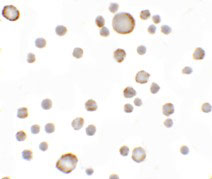
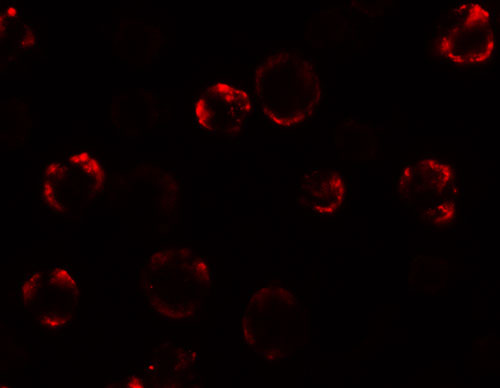
Anosmin Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IF, ICC, E |
|---|---|
| Primary Accession | P23352 |
| Other Accession | NP_000207, 119395746 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
| Calculated MW | 76112 Da |
| Application Notes | Anosmin antibody can be used for detection of Anosmin by Western blot at 1 µg/mL. Antibody can also be used for immunocytochemistry starting at 5 µg/mL. For immunofluorescence start at 20 µg/mL. |
| Gene ID | 3730 |
|---|---|
| Target/Specificity | KAL1; |
| Reconstitution & Storage | Anosmin antibody can be stored at 4 ℃, stable for one year. As with all antibodies care should be taken to avoid repeated freeze thaw cycles. Antibodies should not be exposed to prolonged high temperatures. |
| Precautions | Anosmin Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | ANOS1 (HGNC:6211) |
|---|---|
| Synonyms | ADMLX, KAL, KAL1, KALIG1 |
| Function | Has a dual branch-promoting and guidance activity, which may play an important role in the patterning of mitral and tufted cell collaterals to the olfactory cortex (By similarity). Chemoattractant for fetal olfactory epithelial cells. |
| Cellular Location | Cell membrane; Peripheral membrane protein. Secreted. Note=Proteolytic cleavage may release it from the cell surface into the extracellular space |
| Tissue Location | Expressed in the cerebellum (at protein level). |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Anosmin Antibody: Mutations in Anosmin-1, an extracellular matrix-associated glycosylated protein, have been linked with Kallmann Syndrome (KS), an X-linked genetic disorder characterized by loss of smell caused by abnormal olfactory bulb development and delayed puberty caused by disrupted migration of the gonadotropin-releasing hormone neuron. Anosmin-1 has been shown to directly bind FGFR1 via its N-terminal cysteine-rich domain, whey-acidic protein-like domain, and its first FnIII repeat with the D2 and D3 ectodomains of FGFR1. It is thought that Anosmin-1 can modulate FGFR1 signaling and have opposing effects on the formation and activation of FGF2-FGFR1-heparing complex.
References
Franco B, Guioli S, Pragliola A, et al. A gene deleted in Kallmann’s syndrome shares homology with neural cell adhesion and axonal path-finding molecules. Nature 1991; 353:529-36.
Soussi-Yanicostas N, Hardelin JP, Arroyo-Jimenez MM, et al. Initial characterization of anosmin-1, a putative extracellular matrix protein synthesized by definite neuronal cell populations in the central nervous system. J. Cell Sci. 1996; 109:1749-57.
Hu Y, Guimond SE, Travers P, et al. Novel mechanisms of fibroblast growth factor receptor 1 regulation by extracellular matrix protein Anosmin-1. J. Biol. Chem. 2009; 284:29905-20
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.