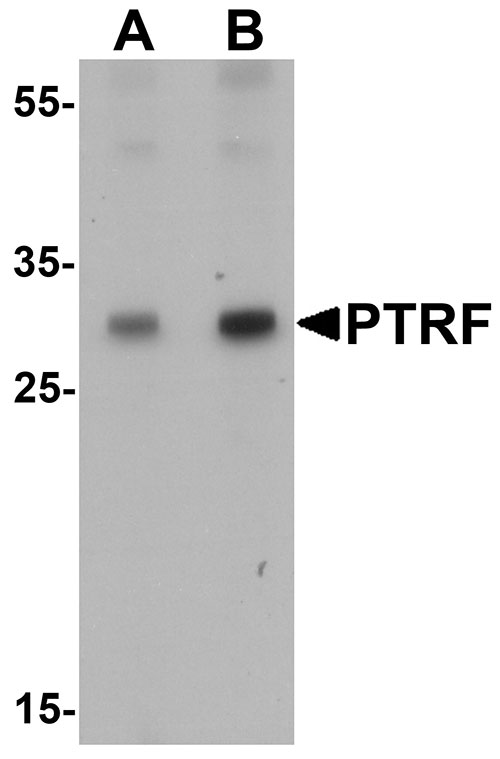
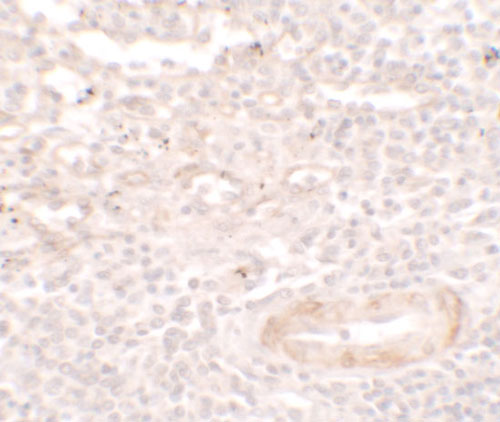
SDPR Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC-P, IF, E |
|---|---|
| Primary Accession | O95810 |
| Other Accession | NP_004648, 4759082 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
| Calculated MW | 47 kDa |
| Application Notes | SDPR antibody can be used for detection of SDPR by Western blot at 1 - 2 µg/mL. |
| Gene ID | 8436 |
|---|---|
| Target/Specificity | SDPR; It is predicted to not cross-react with other members of the cavin family. |
| Reconstitution & Storage | SDPR antibody can be stored at 4℃ for three months and -20℃, stable for up to one year. |
| Precautions | SDPR Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | CAVIN2 (HGNC:10690) |
|---|---|
| Function | Plays an important role in caveolar biogenesis and morphology. Regulates caveolae morphology by inducing membrane curvature within caveolae (PubMed:19525939). Plays a role in caveola formation in a tissue-specific manner. Required for the formation of caveolae in the lung and fat endothelia but not in the heart endothelia. Negatively regulates the size or stability of CAVIN complexes in the lung endothelial cells. May play a role in targeting PRKCA to caveolae (By similarity). |
| Cellular Location | Cytoplasm, cytosol. Membrane, caveola Note=Localizes in the caveolae in a caveolin-dependent manner |
| Tissue Location | Highly expressed in heart and lung, and expressed at lower levels in brain, kidney, liver, pancreas, placenta, and skeletal muscle. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
SDPR Antibody: The serum deprivation-response protein (SDPR) is a calcium-independent phospholipid-binding protein whose expression is increased in serum-starved cells. SDPR is a substrate for protein kinase C (PKC) phosphorylation and recruits the polymerase I and transcript release factor (PTRF) to caveolae. Removal of this protein causes caveolae loss and its over-expression results in caveolae deformation and membrane tubulation. Both SDPR and PTRF, as well as the other member of the cavin family PRKCDBP were down regulated in breast cancer cell lines and breast tumor tissue, suggesting that expression of the cavin family proteins could be a useful prognostic indicator of breast cancer progression.
References
Gustincich S, Vatta P, Goruppi S, et al. The human serum deprivation response gene (SDPR) maps to 2q32-q33 and codes for a phosphatidylserine-binding protein. Genomics 1999; 57:120-9.
Hansen CG, Bright NA, Howard G, et al. SDPR induces membrane curvature and functions in the formation of caveolae. Nat. Cell Biol. 2009; 11:807-14.
Bai L, Deng X, Li Q, et al. Down-regulation of the cavin family proteins in breast cancer. J. Cell Biochem. 2012; 113:322-8.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.