NCBP1 Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, ICC, E |
|---|---|
| Primary Accession | Q09161 |
| Other Accession | NP_002477, 4505343 |
| Reactivity | Human, Mouse, Rat |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
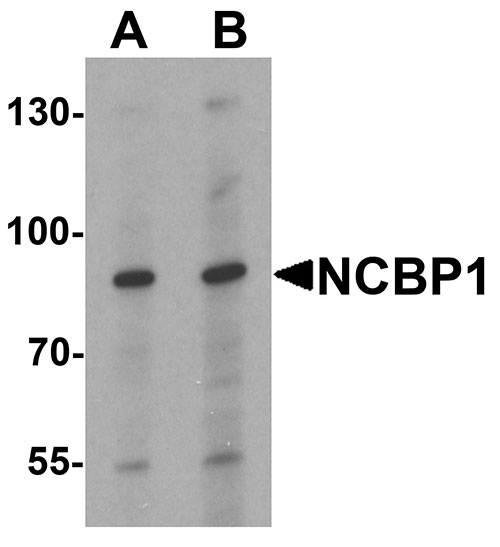
| Calculated MW | Predicted: 87 kDa Observed: 87 kDa |
| Application Notes | NCBP1 antibody can be used for detection of NCBP1 by Western blot at 1 - 2 µg/mL. |
| Gene ID | 4686 |
|---|---|
| Target/Specificity | NCBP1; NCBP1 antibody is human, mouse and rat reactive. At least two isoforms of NCBP1 are known to exist; this antibody will detect both isoforms of NCBP1. |
| Reconstitution & Storage | NCBP1 antibody can be stored at 4℃ for three months and -20℃, stable for up to one year. |
| Precautions | NCBP1 Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | NCBP1 |
|---|---|
| Synonyms | CBP80, NCBP |
| Function | Component of the cap-binding complex (CBC), which binds cotranscriptionally to the 5'-cap of pre-mRNAs and is involved in various processes such as pre-mRNA splicing, translation regulation, nonsense-mediated mRNA decay, RNA-mediated gene silencing (RNAi) by microRNAs (miRNAs) and mRNA export. The CBC complex is involved in mRNA export from the nucleus via its interaction with ALYREF/THOC4/ALY, leading to the recruitment of the mRNA export machinery to the 5'-end of mRNA and to mRNA export in a 5' to 3' direction through the nuclear pore. The CBC complex is also involved in mediating U snRNA and intronless mRNAs export from the nucleus. The CBC complex is essential for a pioneer round of mRNA translation, before steady state translation when the CBC complex is replaced by cytoplasmic cap-binding protein eIF4E. The pioneer round of mRNA translation mediated by the CBC complex plays a central role in nonsense-mediated mRNA decay (NMD), NMD only taking place in mRNAs bound to the CBC complex, but not on eIF4E-bound mRNAs. The CBC complex enhances NMD in mRNAs containing at least one exon-junction complex (EJC) via its interaction with UPF1, promoting the interaction between UPF1 and UPF2. The CBC complex is also involved in 'failsafe' NMD, which is independent of the EJC complex, while it does not participate in Staufen-mediated mRNA decay (SMD). During cell proliferation, the CBC complex is also involved in microRNAs (miRNAs) biogenesis via its interaction with SRRT/ARS2 and is required for miRNA-mediated RNA interference. The CBC complex also acts as a negative regulator of PARN, thereby acting as an inhibitor of mRNA deadenylation. In the CBC complex, NCBP1/CBP80 does not bind directly capped RNAs (m7GpppG-capped RNA) but is required to stabilize the movement of the N-terminal loop of NCBP2/CBP20 and lock the CBC into a high affinity cap-binding state with the cap structure. Associates with NCBP3 to form an alternative cap-binding complex (CBC) which plays a key role in mRNA export and is particularly important in cellular stress situations such as virus infections. The conventional CBC with NCBP2 binds both small nuclear RNA (snRNA) and messenger (mRNA) and is involved in their export from the nucleus whereas the alternative CBC with NCBP3 does not bind snRNA and associates only with mRNA thereby playing a role only in mRNA export. NCBP1/CBP80 is required for cell growth and viability (PubMed:26382858). |
| Cellular Location | Nucleus. Cytoplasm. Note=Localized in cytoplasmic mRNP granules containing untranslated mRNAs. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
NCBP1 Antibody: NCBP1, also known as CBP80, is a component of the nuclear cap-binding protein complex (CBC), which binds to the monomethylated 5' cap of nascent pre-mRNA in the nucleoplasm. NCBP1 promotes high-affinity mRNA-cap binding and associates with the CTD of RNA polymerase II. The CBC promotes pre-mRNA splicing, 3'-end processing, RNA nuclear export, and nonsense-mediated mRNA decay (1,2). Recent evidence has shown that cellular-cap-binding proteins such as NCBP1 associate with influenza virus mRNAs, suggesting that these viral mRNAs may follow the normal cellular pathways for splicing, nuclear export, and translation (3).
References
Ohno M, Kataoka N, and Shimura Y. A nuclear cap binding protein from HeLa cells. Nuc. Acid Res. 1990; 18:6989-95.
Maquat LE, Hwang J, Sato H, et al. CBP80-promoted mRNP rearrangements during the pioneer round of translation, nonsense-mediated mRNA decay, and thereafter. Cold Spring Harbor Symp. Quant. Biol. 2010; 75:127-34.
Bier K, York A, and Fodor E. Cellular cap-binding proteins associate with influenza virus mRNAs. J. Gen. Virol. 92:1627-34.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.