SRPK1 Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC, E |
|---|---|
| Primary Accession | Q96SB4 |
| Other Accession | NP_003128, 47419936 |
| Reactivity | Human, Mouse, Rat |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
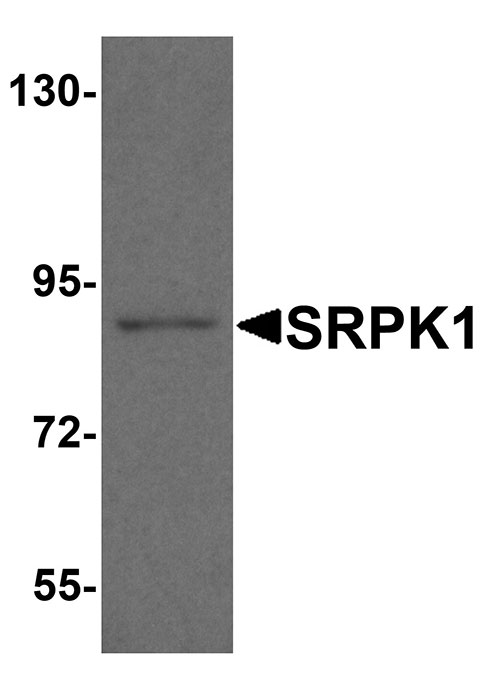
| Calculated MW | Predicted: 54, 72, 91 kDa Observed: 91 kDa |
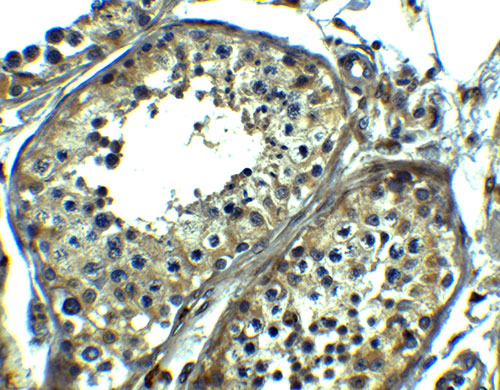
| Application Notes | SRPK1 antibody can be used for the detection of SRPK1 by Western blot at 1 - 2 µg/mL. Antibody can also be used for immunohistochemistry at 5 µg/ml. |
| Gene ID | 6732 |
|---|---|
| Target/Specificity | SRPK1; SRPK1 antibody is human, mouse and rat reactive. Multiple isoforms of SRPK1 are known to exist; this antibody will detect all except isoforms b and e. SRPK1 antibody is predicted to not cross-react with SRPK2. |
| Reconstitution & Storage | SRPK1 antibody can be stored at 4℃ for three months and -20℃, stable for up to one year. |
| Precautions | SRPK1 Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | SRPK1 {ECO:0000312|EMBL:CAC39299.1} |
|---|---|
| Function | Serine/arginine-rich protein-specific kinase which specifically phosphorylates its substrates at serine residues located in regions rich in arginine/serine dipeptides, known as RS domains and is involved in the phosphorylation of SR splicing factors and the regulation of splicing. Plays a central role in the regulatory network for splicing, controlling the intranuclear distribution of splicing factors in interphase cells and the reorganization of nuclear speckles during mitosis. Can influence additional steps of mRNA maturation, as well as other cellular activities, such as chromatin reorganization in somatic and sperm cells and cell cycle progression. Isoform 2 phosphorylates SFRS2, ZRSR2, LBR and PRM1. Isoform 2 phosphorylates SRSF1 using a directional (C-terminal to N-terminal) and a dual-track mechanism incorporating both processive phosphorylation (in which the kinase stays attached to the substrate after each round of phosphorylation) and distributive phosphorylation steps (in which the kinase and substrate dissociate after each phosphorylation event). The RS domain of SRSF1 binds first to a docking groove in the large lobe of the kinase domain of SRPK1. This induces certain structural changes in SRPK1 and/or RRM2 domain of SRSF1, allowing RRM2 to bind the kinase and initiate phosphorylation. The cycles continue for several phosphorylation steps in a processive manner (steps 1-8) until the last few phosphorylation steps (approximately steps 9-12). During that time, a mechanical stress induces the unfolding of the beta-4 motif in RRM2, which then docks at the docking groove of SRPK1. This also signals RRM2 to begin to dissociate, which facilitates SRSF1 dissociation after phosphorylation is completed. Isoform 2 can mediate hepatitis B virus (HBV) core protein phosphorylation. It plays a negative role in the regulation of HBV replication through a mechanism not involving the phosphorylation of the core protein but by reducing the packaging efficiency of the pregenomic RNA (pgRNA) without affecting the formation of the viral core particles. Isoform 1 and isoform 2 can induce splicing of exon 10 in MAPT/TAU. The ratio of isoform 1/isoform 2 plays a decisive role in determining cell fate in K-562 leukaemic cell line: isoform 2 favors proliferation where as isoform 1 favors differentiation. |
| Cellular Location | [Isoform 2]: Cytoplasm. Nucleus. Nucleus matrix. Microsome. Note=Shuttles between the nucleus and the cytoplasm Inhibition of the Hsp90 ATPase activity, osmotic stress and interaction with HHV-1 ICP27 protein can induce its translocation to the nucleus KAT5/TIP60 inhibits its nuclear translocation Cytoplasm. Nucleus, nucleoplasm. Nucleus speckle. Chromosome. Note=Preferentially localizes to the promoter of gene coding regions. |
| Tissue Location | Isoform 2 is predominantly expressed in the testis but is also present at lower levels in heart, ovary, small intestine, liver, kidney, pancreas and skeletal muscle. Isoform 1 is only seen in the testis, at lower levels than isoform 2. Highly expressed in different erythroid and lymphoid cell lines, with isoform 2 being far more abundant than isoform 1. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
SRPK1 and SRPK2, two Arginine/serine-rich (SR) protein-specific kinases constitute control circuits to regulate both constitutive and alternative pre-mRNA splicing and coordinate splicing with transcription in cells (1). They are highly specific for the phosphorylation of these RS proteins, and highly expressed in testis (1,2). SRPK1 plays a role in the condensation of sperm chromatin and controls the intranuclear distribution of splicing factors in interphase cells and the reorganization of nuclear speckles during mitosis (3,4).
References
Wang HY, Arden KC, Bermingham JR Jr, et al. Localization of serine kinases, SRPK1 (SFRSK1) and SRPK2 (SFRSK2), specific for the SR family of splicing factors in mouse and human chromosomes. Genomics 1999; 57:310-5.
Daub H, Blencke S, Habenberger P, et al. Identification of SRPK1 and SRPK2 as the major cellular protein kinases phosphorylating hepatitis B virus core protein. J. Virol. 2002; 76:8124-37.
Zhong XY, Ding JH, Adams JA, et al. Regulation of SR protein phosphorylation and alternative splicing by modulating kinetic interactions of SRPK1 with molecular chaperones. Genes Dev. 2009; 23:482-95.
Mavrou A, Brakspear K, Hamdollah-Zadeh M, et al. Serine-arginine protein kinase 1 (SRPK1) inhibition as a potential novel targeted therapeutic strategy in prostate cancer. Oncogene 2014 Nov 10. doi: 10.1038/onc.2014.360.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.