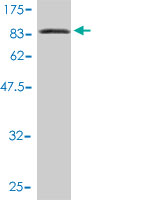
L3MBTL4 Antibody (monoclonal) (M01)
Mouse monoclonal antibody raised against a full length recombinant L3MBTL4.
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB |
|---|---|
| Primary Accession | Q8NA19 |
| Other Accession | BC039316 |
| Reactivity | Human |
| Host | mouse |
| Clonality | Monoclonal |
| Isotype | IgG1 kappa |
| Clone Names | 1B9-1C12 |
| Calculated MW | 71122 Da |
| Gene ID | 91133 |
|---|---|
| Other Names | Lethal(3)malignant brain tumor-like protein 4, H-l(3)mbt-like protein 4, L(3)mbt-like protein 4, L3MBTL4 |
| Target/Specificity | L3MBTL4 (AAH39316, 1 a.a. ~ 534 a.a) full-length recombinant protein with GST tag. MW of the GST tag alone is 26 KDa. |
| Dilution | WB~~1:500~1000 |
| Format | Clear, colorless solution in phosphate buffered saline, pH 7.2 . |
| Storage | Store at -20°C or lower. Aliquot to avoid repeated freezing and thawing. |
| Precautions | L3MBTL4 Antibody (monoclonal) (M01) is for research use only and not for use in diagnostic or therapeutic procedures. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
References
Variation at the NFATC2 Locus Increases the Risk of Thiazolinedinedione-Induced Edema in the Diabetes REduction Assessment with ramipril and rosiglitazone Medication (DREAM) Study. Bailey SD, et al. Diabetes Care, 2010 Jul 13. PMID 20628086.Personalized smoking cessation: interactions between nicotine dose, dependence and quit-success genotype score. Rose JE, et al. Mol Med, 2010 Jul-Aug. PMID 20379614.Gene-centric association signals for lipids and apolipoproteins identified via the HumanCVD BeadChip. Talmud PJ, et al. Am J Hum Genet, 2009 Nov. PMID 19913121.Complete sequencing and characterization of 21,243 full-length human cDNAs. Ota T, et al. Nat Genet, 2004 Jan. PMID 14702039.Generation and initial analysis of more than 15,000 full-length human and mouse cDNA sequences. Strausberg RL, et al. Proc Natl Acad Sci U S A, 2002 Dec 24. PMID 12477932.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.