SLIGKV-NH2 Protein
A Potent Agonist of PAR2, Derived from PAR2 Auto-Ligand Sequence
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND
| Storage | -20°C |
|---|---|
| Precautions | SLIGKV-NH2 Protein is for research use only and not for use in diagnostic or therapeutic procedures. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
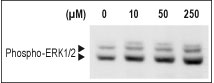
Protease-activated receptors (PARs) 1-4 belong to the superfamily of G-protein-coupled receptors (GPCRs) that are self-activated by a tethered sequence exposed by proteolysis of an extracellular domain1. PARs are activated by proteolysis in response to endogenous and exogenous proteases and can contribute to both cellular homeostasis and pathology1-2. In the case of PAR2, the exposed human peptide SLIGVK-NH2 remains tethered on the receptor and activates a primary binding site located on second loop of the receptor1. As an obvious consequence of its activation mechanism, PAR2 is associated with pathologies with a strong protease release. The involvement of PAR2 in inflammatory diseases such as arthritis, lung inflammation (asthma), inflammatory bowel disease, sepsis, and pain disorders4-6makes PAR2 an attractive target for drug intervention3. The extracellular N-terminus of PAR2 (46 residues long) contains a putative trypsin cleavage site R34 and S351, followed by LIGKV. The human-derived SLIGKV-NH2is a small peptide that mimics the ligand binding properties of the tethered ligand exposed by proteolysis of the N-terminus from the natural receptor7 and hence, a significant tool used to study PAR2.
References
1 . Ossovskaya VS. et al.(2004)Physiol Rev.84,579.
2 . Rattenholl A. et al.(2008)Drug News Perspect. 21,369.
3 . Boitano S. et al.(2011)J. Med. Chem.54,1308.
4 . Barry GD. et al. (2006) Curr. Med. Chem.13,243.
5 . Steinhoff M. et al. (2005) Endocr.Rev.26, 1.
6 . Cottrell GS. et al.(2003)Biochem. Soc.Trans. 31, 1191.
7 . Ramachandran R. et al.(2008)Br. J. Pharmacol.153,S263.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.