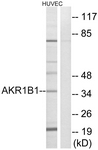
AKR1B1 / Aldose Reductase Antibody (aa241-290)
Rabbit Polyclonal Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC-P, E |
|---|---|
| Primary Accession | P15121 |
| Other Accession | 231 |
| Reactivity | Human, Rat |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
| Calculated MW | 35853 Da |
| Dilution | ELISA (1:20000), IHC-P (20 µg/ml), WB (1:500 - 1:1000), |
| Gene ID | 231 |
|---|---|
| Other Names | AKR1B1, Aldose reductase, ALDR1, ADR, Aldehyde reductase, Aldehyde reductase 1, ALR2, Lii5-2 CTCL tumor antigen, Low Km aldose reductase |
| Target/Specificity | AKR1B1 Antibody detects endogenous levels of total AKR1B1 protein. |
| Reconstitution & Storage | PBS (without Mg2+, Ca2+), pH 7.4, 150 mM sodium chloride, 0.02% sodium azide, 50% glycerol. Store at -20°C for up to one year. |
| Precautions | AKR1B1 / Aldose Reductase Antibody (aa241-290) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | AKR1B1 |
|---|---|
| Synonyms | ALDR1, ALR2 {ECO:0000303|PubMed:17368668 |
| Function | Catalyzes the NADPH-dependent reduction of a wide variety of carbonyl-containing compounds to their corresponding alcohols. Displays enzymatic activity towards endogenous metabolites such as aromatic and aliphatic aldehydes, ketones, monosacharides, bile acids and xenobiotics substrates. Key enzyme in the polyol pathway, catalyzes reduction of glucose to sorbitol during hyperglycemia (PubMed:1936586). Reduces steroids and their derivatives and prostaglandins. Displays low enzymatic activity toward all-trans-retinal, 9-cis-retinal, and 13-cis- retinal (PubMed:12732097, PubMed:19010934, PubMed:8343525). Catalyzes the reduction of diverse phospholipid aldehydes such as 1-palmitoyl-2- (5-oxovaleroyl)-sn -glycero-3-phosphoethanolamin (POVPC) and related phospholipid aldehydes that are generated from the oxydation of phosphotidylcholine and phosphatdyleethanolamides (PubMed:17381426). Plays a role in detoxifying dietary and lipid-derived unsaturated carbonyls, such as crotonaldehyde, 4-hydroxynonenal, trans-2-hexenal, trans-2,4-hexadienal and their glutathione-conjugates carbonyls (GS- carbonyls) (PubMed:21329684). |
| Cellular Location | Cytoplasm. |
| Tissue Location | Highly expressed in embryonic epithelial cells (EUE) in response to osmotic stress. |
| Volume | 50 µl |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Catalyzes the NADPH-dependent reduction of a wide variety of carbonyl-containing compounds to their corresponding alcohols with a broad range of catalytic efficiencies.
References
Bohren K.M.,et al.J. Biol. Chem. 264:9547-9551(1989).
Chung S.,et al.J. Biol. Chem. 264:14775-14777(1989).
Graham A.,et al.Nucleic Acids Res. 17:8368-8368(1989).
Grundmann U.,et al.DNA Cell Biol. 9:149-157(1990).
Nishimura C.,et al.J. Biol. Chem. 265:9788-9792(1990).
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.