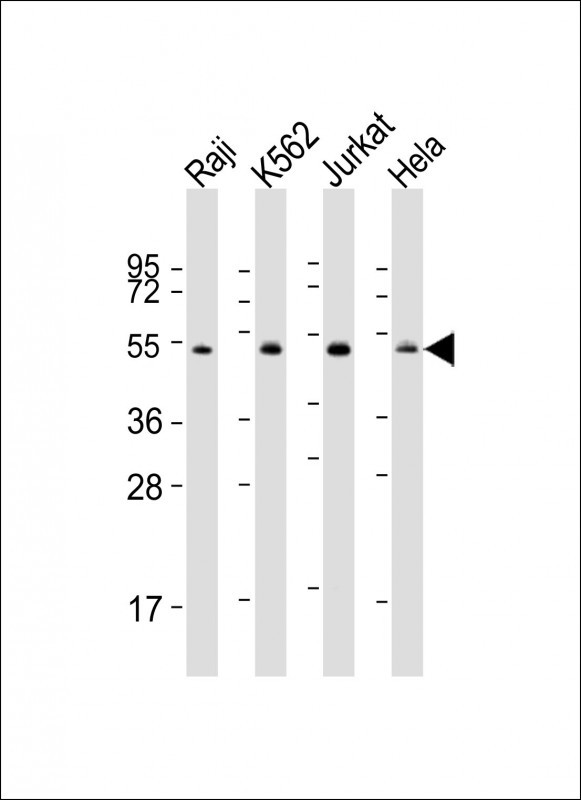
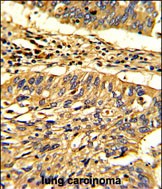
Hsp 60 Antibody (N-term)
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC-P, E |
|---|---|
| Primary Accession | Q0VDF9 |
| Other Accession | Q99M31 |
| Reactivity | Human, Mouse |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 54794 Da |
| Antigen Region | 80-109 aa |
| Gene ID | 51182 |
|---|---|
| Other Names | Heat shock 70 kDa protein 14, HSP70-like protein 1, Heat shock protein HSP60, HSPA14, HSP60, HSP70L1 |
| Target/Specificity | This Hsp 60 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 80-109 amino acids from the N-terminal region of human Hsp 60. |
| Dilution | WB~~1:1000 IHC-P~~1:25 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | Hsp 60 Antibody (N-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | HSPA14 |
|---|---|
| Synonyms | HSP60, HSP70L1 |
| Function | Component of the ribosome-associated complex (RAC), a complex involved in folding or maintaining nascent polypeptides in a folding- competent state. In the RAC complex, binds to the nascent polypeptide chain, while DNAJC2 stimulates its ATPase activity. |
| Cellular Location | Cytoplasm, cytosol. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Hsp60 is a member of a highly conserved family which includes molecular chaperones from several species such as plant Hsp60 (known as Rubisco binding protein), GroEL, the E.coli Hsp60 and 65 kDa major antigen of mycobacteria. In eukaryotes, Hsp60 is localized in the mitochondrial matrix and in plants Hsp60 is localized in the chloroplast. Mitochondria, chloroplasts and bacteria have a common ancestry (>1 billion years) and this fact together with the high degree of homology between the divegent Hsp60s would indicate that these proteins carry out a primitive but important function which is similar to all of these different species.The common characteristics of the Hsp60s from the divergent species are i) high abundance, ii) induction with environmental stress such as heat shock, iii) homo oligomeric structures of either 7 or 14 subunits which reversibly dissociate in the presence of magnesium ions and ATP, iv) ATPase activity and v) a role in folding and assembly of oligomeric protein structures. These similarities are supported by recent studies where the single ring human mitochondrial homolog, Hsp60 with its co chaperonin, Hsp10 were expressed in a E. coli strain, engineered so that the groE operon is under strict regulatory control. This study has demonstrated that expression of Hsp60-Hsp10 was able to carry out all essential in vivo functions of GroEL and its co chaperonin, GroES. Consistent with their functions as chaperones, Hsp60 and Hsp10 have been suggested to act as docking molecules with a passive role in the maturation of caspase processing. Data demonstrates that recombinant Hsp60 and Hsp10 have been shown to accelerate the activation of procaspase 3 by cytochrome c and dATP in an ATP dependent manner. Hsps are intracellular proteins which are thought to serve protective functions against infection and cellular stress, however several recent studies indicate that members of the Hsp60 family are linked to a number of autoimmune diseases, artherosclerosis and chlamydial disease.
References
Velez,D.R., et.al., Am. J. Obstet. Gynecol. 200 (2), 209 (2009)
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.