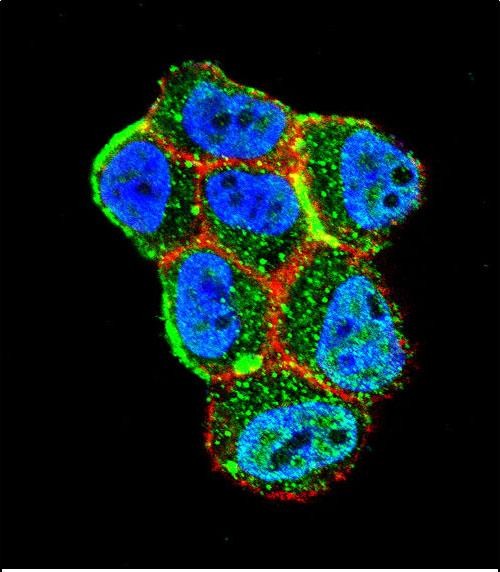
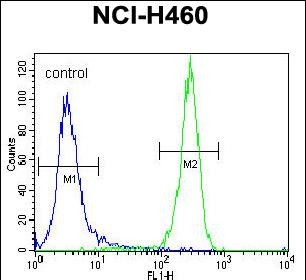
STK11 (LKB1) Antibody (N-term I29)
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS: 2
- PROTOCOLS
- BACKGROUND

Application
| IF, FC, WB, IHC-P, E |
|---|---|
| Primary Accession | Q15831 |
| Other Accession | Q91604, D4AE59, Q9WTK7, Q0GGW5 |
| Reactivity | Human |
| Predicted | Chicken, Mouse, Rat, Xenopus |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 48636 Da |
| Antigen Region | 14-44 aa |
| Gene ID | 6794 |
|---|---|
| Other Names | Serine/threonine-protein kinase STK11, Liver kinase B1, LKB1, hLKB1, Renal carcinoma antigen NY-REN-19, STK11, LKB1, PJS |
| Target/Specificity | This STK11 (LKB1) antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 14-44 amino acids from the N-terminal region of human STK11 (LKB1). |
| Dilution | IF~~1:10~50 WB~~1:1000 IHC-P~~1:50~100 FC~~1:10~50 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis against PBS. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | STK11 (LKB1) Antibody (N-term I29) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | STK11 (HGNC:11389) |
|---|---|
| Synonyms | LKB1, PJS |
| Function | Tumor suppressor serine/threonine-protein kinase that controls the activity of AMP-activated protein kinase (AMPK) family members, thereby playing a role in various processes such as cell metabolism, cell polarity, apoptosis and DNA damage response. Acts by phosphorylating the T-loop of AMPK family proteins, thus promoting their activity: phosphorylates PRKAA1, PRKAA2, BRSK1, BRSK2, MARK1, MARK2, MARK3, MARK4, NUAK1, NUAK2, SIK1, SIK2, SIK3 and SNRK but not MELK. Also phosphorylates non-AMPK family proteins such as STRADA, PTEN and possibly p53/TP53. Acts as a key upstream regulator of AMPK by mediating phosphorylation and activation of AMPK catalytic subunits PRKAA1 and PRKAA2 and thereby regulates processes including: inhibition of signaling pathways that promote cell growth and proliferation when energy levels are low, glucose homeostasis in liver, activation of autophagy when cells undergo nutrient deprivation, and B-cell differentiation in the germinal center in response to DNA damage. Also acts as a regulator of cellular polarity by remodeling the actin cytoskeleton. Required for cortical neuron polarization by mediating phosphorylation and activation of BRSK1 and BRSK2, leading to axon initiation and specification. Involved in DNA damage response: interacts with p53/TP53 and recruited to the CDKN1A/WAF1 promoter to participate in transcription activation. Able to phosphorylate p53/TP53; the relevance of such result in vivo is however unclear and phosphorylation may be indirect and mediated by downstream STK11/LKB1 kinase NUAK1. Also acts as a mediator of p53/TP53-dependent apoptosis via interaction with p53/TP53: translocates to the mitochondrion during apoptosis and regulates p53/TP53-dependent apoptosis pathways. Regulates UV radiation-induced DNA damage response mediated by CDKN1A. In association with NUAK1, phosphorylates CDKN1A in response to UV radiation and contributes to its degradation which is necessary for optimal DNA repair (PubMed:25329316). |
| Cellular Location | Nucleus. Cytoplasm. Membrane. Mitochondrion. Note=A small fraction localizes at membranes (By similarity). Relocates to the cytoplasm when bound to STRAD (STRADA or STRADB) and CAB39/MO25 (CAB39/MO25alpha or CAB39L/MO25beta) Translocates to the mitochondrion during apoptosis. PTEN promotes cytoplasmic localization. |
| Tissue Location | Ubiquitously expressed. Strongest expression in testis and fetal liver |

Provided below are standard protocols that you may find useful for product applications.
Background
STK11, is a member of the serine/threonine kinase family, regulates cell polarity and functions as a tumor suppressor. Mutations in STK11 have been associated with Peutz-Jeghers syndrome, an autosomal dominant disorder characterized by the growth of polyps in the gastrointestinal tract, pigmented macules on the skin and mouth, and other neoplasms.
References
Mart, et al., J. Biotechnol. 115(1):23-34 (2005).
Shaw, R.J., et al., Proc. Natl. Acad. Sci. U.S.A. 101(10):3329-3335 (2004).
Suzuki, A., et al., Biochem. Biophys. Res. Commun. 324(3):986-992 (2004).
Hearle, N., et al., Genes Chromosomes Cancer 41(2):163-169 (2004).
Corradetti, M.N., et al., Genes Dev. 18(13):1533-1538 (2004).
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.














 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.