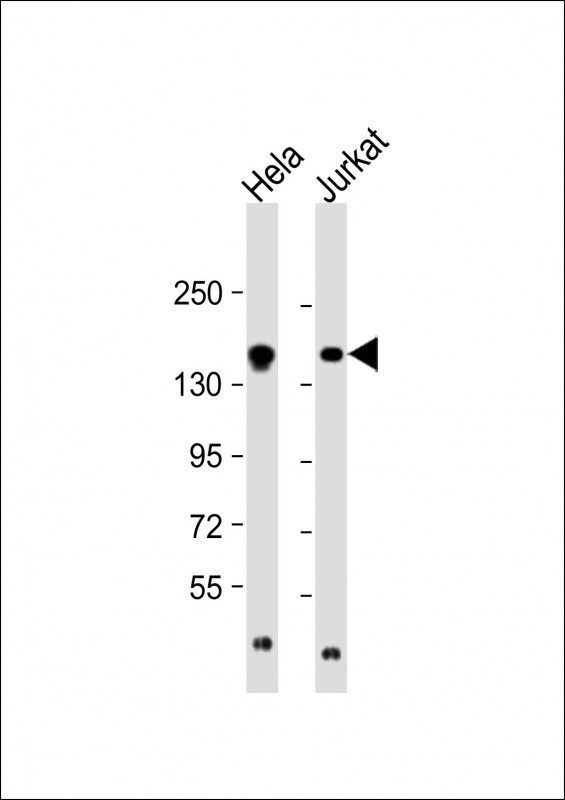
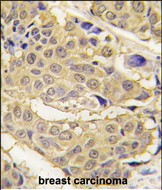
EPRS Antibody (N-term)
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS: 1
- PROTOCOLS
- BACKGROUND

Application
| IHC-P, WB, E |
|---|---|
| Primary Accession | P07814 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 170591 Da |
| Antigen Region | 285-314 aa |
| Gene ID | 2058 |
|---|---|
| Other Names | Bifunctional glutamate/proline--tRNA ligase, Bifunctional aminoacyl-tRNA synthetase, Cell proliferation-inducing gene 32 protein, Glutamatyl-prolyl-tRNA synthetase, Glutamate--tRNA ligase, Glutamyl-tRNA synthetase, GluRS, Proline--tRNA ligase, Prolyl-tRNA synthetase, EPRS, GLNS, PARS, QARS, QPRS |
| Target/Specificity | This EPRS antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 285-314 amino acids from the N-terminal region of human EPRS. |
| Dilution | WB~~1:8000 IHC-P~~1:10~50 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis against PBS. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | EPRS Antibody (N-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | EPRS1 (HGNC:3418) |
|---|---|
| Function | Multifunctional protein which primarily functions within the aminoacyl-tRNA synthetase multienzyme complex, also known as multisynthetase complex. Within the complex it catalyzes the attachment of both L-glutamate and L-proline to their cognate tRNAs in a two-step reaction where the amino acid is first activated by ATP to form a covalent intermediate with AMP. Subsequently, the activated amino acid is transferred to the acceptor end of the cognate tRNA to form L- glutamyl-tRNA(Glu) and L-prolyl-tRNA(Pro) (PubMed:3290852, PubMed:29576217, PubMed:24100331, PubMed:23263184, PubMed:37212275). Upon interferon-gamma stimulation, EPRS1 undergoes phosphorylation, causing its dissociation from the aminoacyl-tRNA synthetase multienzyme complex. It is recruited to form the GAIT complex, which binds to stem loop-containing GAIT elements found in the 3'-UTR of various inflammatory mRNAs, such as ceruloplasmin. The GAIT complex inhibits the translation of these mRNAs, allowing interferon-gamma to redirect the function of EPRS1 from protein synthesis to translation inhibition in specific cell contexts (PubMed:15479637, PubMed:23071094). Furthermore, it can function as a downstream effector in the mTORC1 signaling pathway, by promoting the translocation of SLC27A1 from the cytoplasm to the plasma membrane where it mediates the uptake of long- chain fatty acid by adipocytes. Thereby, EPRS1 also plays a role in fat metabolism and more indirectly influences lifespan (PubMed:28178239). |
| Cellular Location | Cytoplasm, cytosol. Membrane; Peripheral membrane protein Note=Translocates from cytosol to membranes upon phosphorylation at Ser-999. |

Provided below are standard protocols that you may find useful for product applications.
Background
Aminoacyl-tRNA synthetases are a class of enzymes that charge tRNAs with their cognate amino acids. EPRS is a multifunctional aminoacyl-tRNA synthetase that catalyzes the aminoacylation of glutamic acid and proline tRNA species.
References
Jia,J., Mol. Cell 29 (6), 679-690 (2008)
Beausoleil,S.A., Nat. Biotechnol. 24 (10), 1285-1292 (2006)
Kato,T., Cancer Res. 65 (13), 5638-5646 (2005)
Sampath,P., Cell 119 (2), 195-208 (2004)
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.















 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.