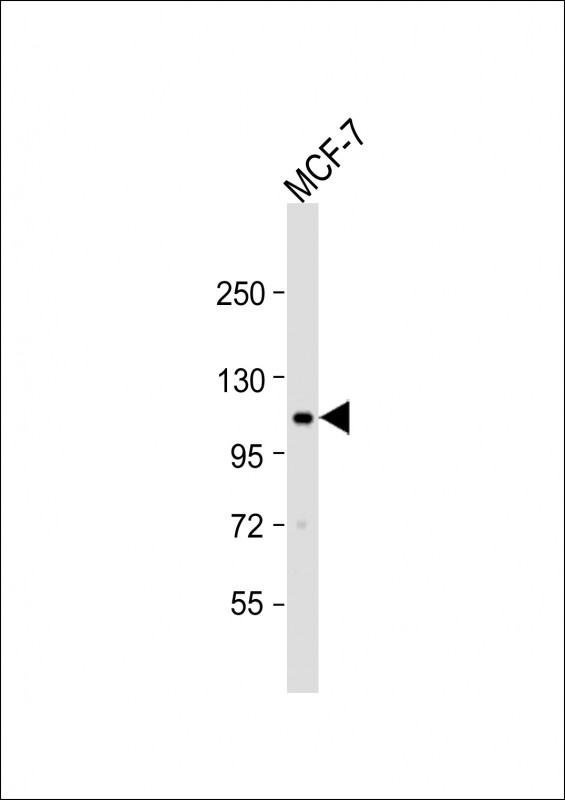
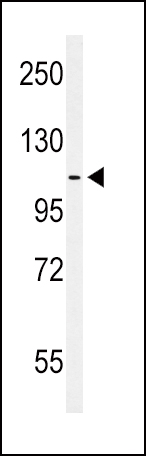
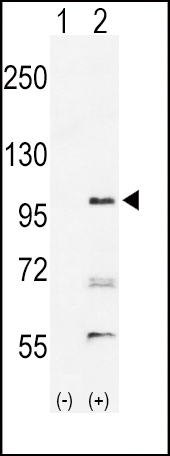
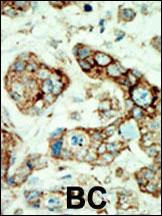
EphA2 Antibody (N-term)
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS: 3
- PROTOCOLS
- BACKGROUND

Application
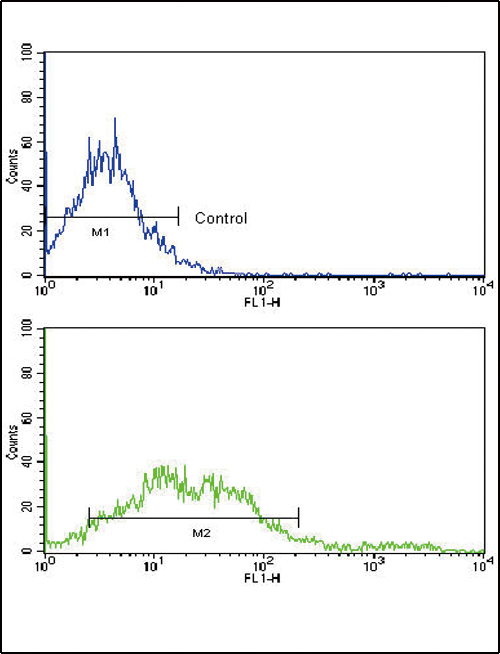
| IHC-P, FC, WB, E |
|---|---|
| Primary Accession | P29317 |
| Other Accession | Q1KL86 |
| Reactivity | Human, Mouse |
| Predicted | Monkey |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 108266 Da |
| Antigen Region | 30-60 aa |
| Gene ID | 1969 |
|---|---|
| Other Names | Ephrin type-A receptor 2, Epithelial cell kinase, Tyrosine-protein kinase receptor ECK, EPHA2, ECK |
| Target/Specificity | This EphA2 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 30-60 amino acids from the N-terminal region of human EphA2. |
| Dilution | WB~~1:1000 IHC-P~~1:50~100 FC~~1:10~50 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis against PBS. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | EphA2 Antibody (N-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | EPHA2 |
|---|---|
| Synonyms | ECK |
| Function | Receptor tyrosine kinase which binds promiscuously membrane- bound ephrin-A family ligands residing on adjacent cells, leading to contact-dependent bidirectional signaling into neighboring cells. The signaling pathway downstream of the receptor is referred to as forward signaling while the signaling pathway downstream of the ephrin ligand is referred to as reverse signaling. Activated by the ligand ephrin- A1/EFNA1 regulates migration, integrin-mediated adhesion, proliferation and differentiation of cells. Regulates cell adhesion and differentiation through DSG1/desmoglein-1 and inhibition of the ERK1/ERK2 (MAPK3/MAPK1, respectively) signaling pathway. May also participate in UV radiation-induced apoptosis and have a ligand- independent stimulatory effect on chemotactic cell migration. During development, may function in distinctive aspects of pattern formation and subsequently in development of several fetal tissues. Involved for instance in angiogenesis, in early hindbrain development and epithelial proliferation and branching morphogenesis during mammary gland development. Engaged by the ligand ephrin-A5/EFNA5 may regulate lens fiber cells shape and interactions and be important for lens transparency development and maintenance. With ephrin-A2/EFNA2 may play a role in bone remodeling through regulation of osteoclastogenesis and osteoblastogenesis. |
| Cellular Location | Cell membrane; Single-pass type I membrane protein. Cell projection, ruffle membrane; Single-pass type I membrane protein. Cell projection, lamellipodium membrane; Single-pass type I membrane protein. Cell junction, focal adhesion. Note=Present at regions of cell-cell contacts but also at the leading edge of migrating cells (PubMed:19573808, PubMed:20861311). Relocates from the plasma membrane to the cytoplasmic and perinuclear regions in cancer cells (PubMed:18794797). |
| Tissue Location | Expressed in brain and glioma tissue and glioma cell lines (at protein level). Expressed most highly in tissues that contain a high proportion of epithelial cells, e.g. skin, intestine, lung, and ovary. |

Provided below are standard protocols that you may find useful for product applications.
Background
Protein kinases are enzymes that transfer a phosphate group from a phosphate donor, generally the g phosphate of ATP, onto an acceptor amino acid in a substrate protein. By this basic mechanism, protein kinases mediate most of the signal transduction in eukaryotic cells, regulating cellular metabolism, transcription, cell cycle progression, cytoskeletal rearrangement and cell movement, apoptosis, and differentiation. With more than 500 gene products, the protein kinase family is one of the largest families of proteins in eukaryotes. The family has been classified in 8 major groups based on sequence comparison of their tyrosine (PTK) or serine/threonine (STK) kinase catalytic domains. The tyrosine kinase (TK) group is mainly involved in the regulation of cell-cell interactions such as differentiation, adhesion, motility and death. There are currently about 90 TK genes sequenced, 58 are of receptor protein TK (e.g. EGFR, EPH, FGFR, PDGFR, TRK, and VEGFR families), and 32 of cytosolic TK (e.g. ABL, FAK, JAK, and SRC families).
References
Lindberg, R.A., et al., Mol. Cell. Biol. 10(12):6316-6324 (1990).
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.














 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.