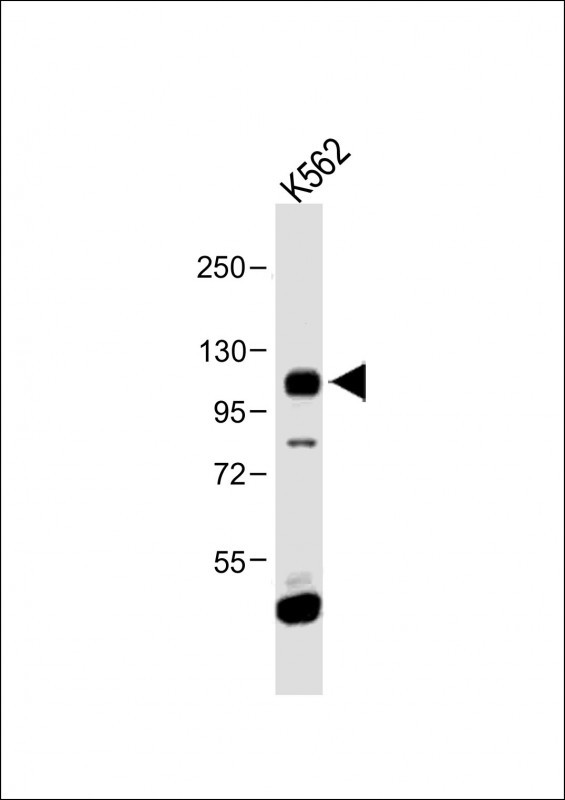
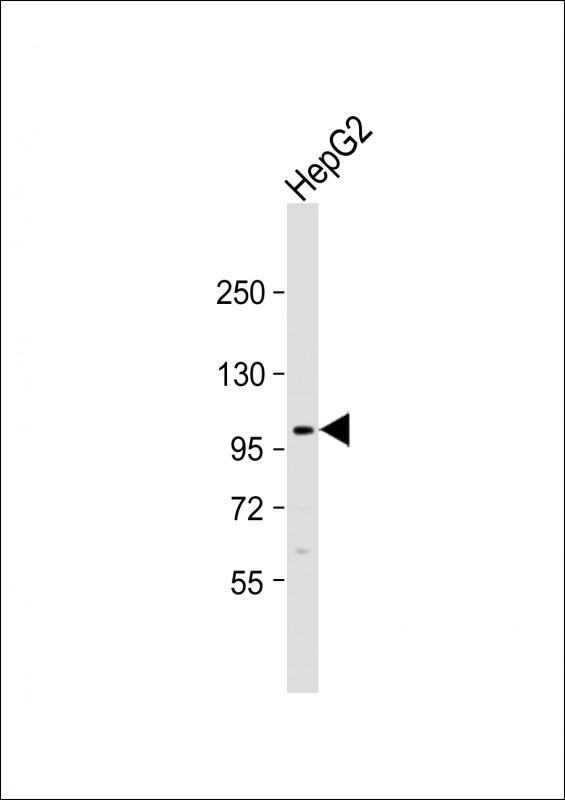
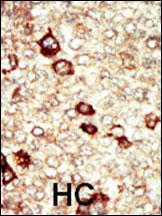
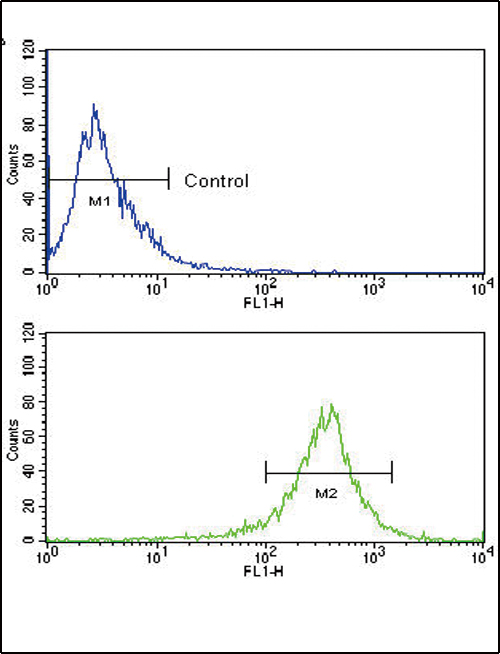
FGFR4 Antibody (N-term)
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| IHC-P, FC, WB, E |
|---|---|
| Primary Accession | P22455 |
| Reactivity | Human, Mouse |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 87954 Da |
| Antigen Region | 24-55 aa |
| Gene ID | 2264 |
|---|---|
| Other Names | Fibroblast growth factor receptor 4, FGFR-4, CD334, FGFR4, JTK2, TKF |
| Target/Specificity | This FGFR4 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 24-55 amino acids from the N-terminal region of human FGFR4. |
| Dilution | WB~~1:1000 IHC-P~~1:50~100 FC~~1:10~50 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis against PBS. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | FGFR4 Antibody (N-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | FGFR4 |
|---|---|
| Synonyms | JTK2, TKF |
| Function | Tyrosine-protein kinase that acts as a cell-surface receptor for fibroblast growth factors and plays a role in the regulation of cell proliferation, differentiation and migration, and in regulation of lipid metabolism, bile acid biosynthesis, glucose uptake, vitamin D metabolism and phosphate homeostasis. Required for normal down- regulation of the expression of CYP7A1, the rate-limiting enzyme in bile acid synthesis, in response to FGF19. Phosphorylates PLCG1 and FRS2. Ligand binding leads to the activation of several signaling cascades. Activation of PLCG1 leads to the production of the cellular signaling molecules diacylglycerol and inositol 1,4,5-trisphosphate. Phosphorylation of FRS2 triggers recruitment of GRB2, GAB1, PIK3R1 and SOS1, and mediates activation of RAS, MAPK1/ERK2, MAPK3/ERK1 and the MAP kinase signaling pathway, as well as of the AKT1 signaling pathway. Promotes SRC-dependent phosphorylation of the matrix protease MMP14 and its lysosomal degradation. FGFR4 signaling is down-regulated by receptor internalization and degradation; MMP14 promotes internalization and degradation of FGFR4. Mutations that lead to constitutive kinase activation or impair normal FGFR4 inactivation lead to aberrant signaling. |
| Cellular Location | Cell membrane; Single-pass type I membrane protein. Endosome. Endoplasmic reticulum. Note=Internalized from the cell membrane to recycling endosomes, and from there back to the cell membrane |
| Tissue Location | Expressed in gastrointestinal epithelial cells, pancreas, and gastric and pancreatic cancer cell lines |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
FGFR4 is a member of the fibroblast growth factor receptor family, where amino acid sequence is highly conserved between members and throughout evolution. FGFR family members differ from one another in their ligand affinities and tissue distribution. A full-length representative protein would consist of an extracellular region, composed of three immunoglobulin-like domains, a single hydrophobic membrane-spanning segment and a cytoplasmic tyrosine kinase domain. The extracellular portion of the protein interacts with fibroblast growth factors, setting in motion a cascade of downstream signals, ultimately influencing mitogenesis and differentiation. The genomic organization of the gene, compared to members 1-3, encompasses 18 exons rather than 19 or 20. Although alternative splicing has been observed, there is no evidence that the C-terminal half of the IgIII domain of this protein varies between three alternate forms, as indicated for members 1-3. This particular family member preferentially binds acidic fibroblast growth factor and, although its specific function is unknown, it is overexpressed in gynecological tumor samples, suggesting a role in breast and ovarian tumorigenesis.
References
References for protein:
1.Jezequel, P., et al., Br. J. Cancer 90(1):189-193 (2004).
2.Qian, Z.R., et al., J. Clin. Endocrinol. Metab. 89(4):1904-1911 (2004).
3.Yu, S., et al., J. Biol. Chem. 278(22):19597-19602 (2003).
4.Becker, N., et al., Cancer Epidemiol. Biomarkers Prev. 12(6):582-583 (2003).
5.Takenaka, H., et al., Arch. Dermatol. Res. 294(7):331-338 (2002).
References for HeLa cell line:
1. Scherer WF, Syverton JT, Gey GO (May 1953). "Studies on the propagation in vitro of poliomyelitis viruses. IV. Viral multiplication in a stable strain of human malignant epithelial cells (strain HeLa) derived from an epidermoid carcinoma of the cervix". J. Exp. Med. 97 (5): 695–710. [PubMed:13052828].
2. Macville M, Schröck E, Padilla-Nash H, Keck C, Ghadimi BM, Zimonjic D, Popescu N, Ried T (January 1999). "Comprehensive and definitive molecular cytogenetic characterization of HeLa cells by spectral karyotyping". Cancer Res. 59 (1): 141–50. [PubMed: 9892199].
3. Rahbari R, Sheahan T, Modes V, Collier P, Macfarlane C, Badge RM (April 2009). "A novel L1 retrotransposon marker for HeLa cell line identification". BioTechniques 46 (4): 277–84. [PubMed: 19450234].
4. Capes-Davis A, Theodosopoulos G, Atkin I, Drexler HG, Kohara A, MacLeod RA, Masters JR, Nakamura Y, Reid YA, Reddel RR, Freshney RI (July 2010). "Check your cultures! A list of cross-contaminated or misidentified cell lines". Int. J. Cancer 127 (1): 1–8. [PubMed:20143388].
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.