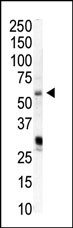
PIP5K1A Antibody (N-term)
Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS: 2
- PROTOCOLS
- BACKGROUND

Application
| WB, E |
|---|---|
| Primary Accession | Q99755 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 62633 Da |
| Antigen Region | 19-50 aa |
| Gene ID | 8394 |
|---|---|
| Other Names | Phosphatidylinositol 4-phosphate 5-kinase type-1 alpha, PIP5K1-alpha, PtdIns(4)P-5-kinase 1 alpha, 68 kDa type I phosphatidylinositol 4-phosphate 5-kinase alpha, Phosphatidylinositol 4-phosphate 5-kinase type I alpha, PIP5KIalpha, PIP5K1A |
| Target/Specificity | This PIP5K1A antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 19-50 amino acids from the N-terminal region of human PIP5K1A. |
| Dilution | WB~~1:1000 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is prepared by Saturated Ammonium Sulfate (SAS) precipitation followed by dialysis against PBS. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | PIP5K1A Antibody (N-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | PIP5K1A (HGNC:8994) |
|---|---|
| Function | Catalyzes the phosphorylation of phosphatidylinositol 4- phosphate (PtdIns(4)P/PI4P) to form phosphatidylinositol 4,5- bisphosphate (PtdIns(4,5)P2/PIP2), a lipid second messenger that regulates several cellular processes such as signal transduction, vesicle trafficking, actin cytoskeleton dynamics, cell adhesion, and cell motility (PubMed:8955136, PubMed:21477596, PubMed:22942276). PtdIns(4,5)P2 can directly act as a second messenger or can be utilized as a precursor to generate other second messengers: inositol 1,4,5- trisphosphate (IP3), diacylglycerol (DAG) or phosphatidylinositol- 3,4,5-trisphosphate (PtdIns(3,4,5)P3/PIP3) (PubMed:19158393, PubMed:20660631). PIP5K1A-mediated phosphorylation of PtdIns(4)P is the predominant pathway for PtdIns(4,5)P2 synthesis (By similarity). Can also use phosphatidylinositol (PtdIns) as substrate in vitro (PubMed:22942276). Together with PIP5K1C, is required for phagocytosis, both enzymes regulating different types of actin remodeling at sequential steps (By similarity). Promotes particle ingestion by activating the WAS GTPase-binding protein that induces Arp2/3 dependent actin polymerization at the nascent phagocytic cup (By similarity). Together with PIP5K1B, is required, after stimulation by G-protein coupled receptors, for the synthesis of IP3 that will induce stable platelet adhesion (By similarity). Recruited to the plasma membrane by the E-cadherin/beta-catenin complex where it provides the substrate PtdIns(4,5)P2 for the production of PtdIns(3,4,5)P3, IP3 and DAG, that will mobilize internal calcium and drive keratinocyte differentiation (PubMed:19158393). Positively regulates insulin-induced translocation of SLC2A4 to the cell membrane in adipocytes (By similarity). Together with PIP5K1C has a role during embryogenesis (By similarity). Independently of its catalytic activity, is required for membrane ruffling formation, actin organization and focal adhesion formation during directional cell migration by controlling integrin-induced translocation of the small GTPase RAC1 to the plasma membrane (PubMed:20660631). Also functions in the nucleus where it acts as an activator of TUT1 adenylyltransferase activity in nuclear speckles, thereby regulating mRNA polyadenylation of a select set of mRNAs (PubMed:18288197). |
| Cellular Location | Cell membrane {ECO:0000250|UniProtKB:P70182}. Cytoplasm {ECO:0000250|UniProtKB:P70182}. Nucleus. Nucleus speckle. Cell projection, ruffle. Cell projection, lamellipodium. Note=Colocalizes with RAC1 at actin-rich membrane ruffles (PubMed:20660631). Localizes to nuclear speckles and associates with TUT1 to regulate polyadenylation of selected mRNAs (PubMed:18288197). |
| Tissue Location | Highly expressed in heart, placenta, skeletal muscle, kidney and pancreas. Detected at lower levels in brain, lung and liver. |

Provided below are standard protocols that you may find useful for product applications.
Background
Overexpression of phosphatidylinositol phosphate 5-kinase alpha (PIP5KIalpha), which synthesizes PIP2, suppresses apoptosis, whereas a kinase-deficient mutant does not. Protection by the wild-type PIP5KIalpha isaccompanied by decreases in the generation of activated caspases and of caspase 3-cleaved PARP. Protection is not mediated through PIP3 or Akt activation. An anti-apoptotic role for PIP(2) is substantiated by the finding that PIP5KIalpha is cleaved by caspase 3 during apoptosis, and cleavage inactivates PIP5KIalpha in vitro. Mutation of the P(4) position (D279A) of the PIP5KIalpha caspase 3 cleavage consensus prevents cleavage in vitro, and during apoptosis in vivo. Significantly, the caspase 3-resistant PIP5KIalpha mutant is more effective in suppressing apoptosis than the wild-type kinase. PIP2 is a direct regulator of apical and effector caspases in the death receptor and mitochondrial pathways, and PIP5KIalpha inactivation contributes to the progression of apoptosis.
References
Doughman, R.L., et al., J. Biol. Chem. 278(25):23036-23045 (2003).
Loijens, J.C., et al., J. Biol. Chem. 271(51):32937-32943 (1996).
Xie, Y., et al., Cytogenet. Cell Genet. 88 (3-4), 197-199 (2000).
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.















 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.