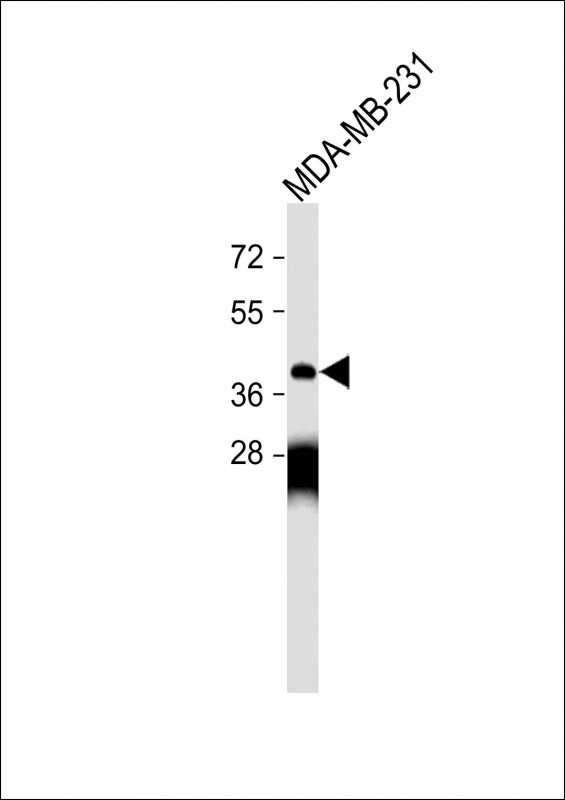
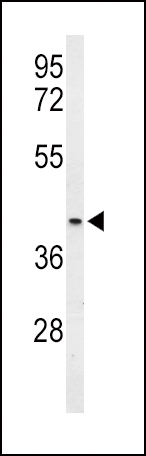
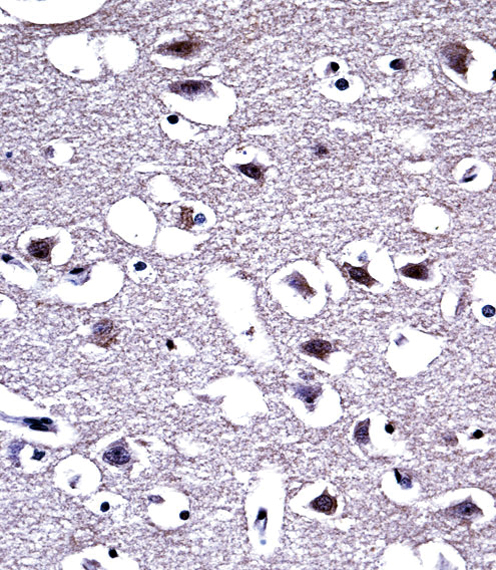
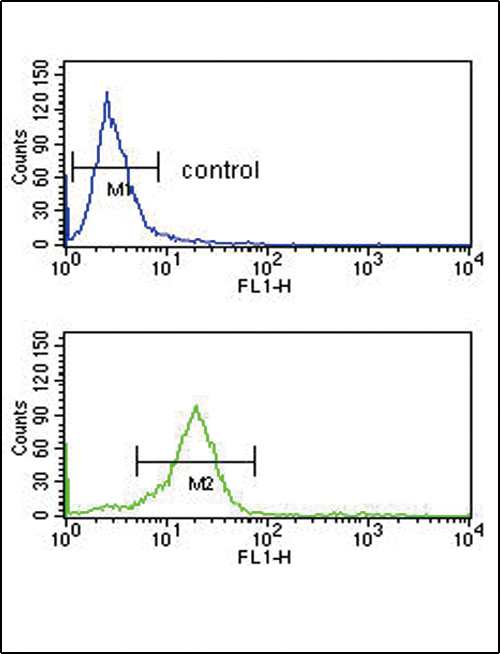
OPRM1 Antibody (Center)
Affinity Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, FC, IHC-P, E |
|---|---|
| Primary Accession | P35372 |
| Other Accession | P33535, Q95247, P42866, Q95M54 |
| Reactivity | Human, Mouse |
| Predicted | Monkey, Pig, Rat |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 44779 Da |
| Antigen Region | 161-187 aa |
| Gene ID | 4988 |
|---|---|
| Other Names | Mu-type opioid receptor, M-OR-1, MOR-1, Mu opiate receptor, Mu opioid receptor, MOP, hMOP, OPRM1, MOR1 |
| Target/Specificity | This OPRM1 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 161-187 amino acids from the Central region of human OPRM1. |
| Dilution | WB~~1:1000 IHC-P~~1:10~50 FC~~1:10~50 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | OPRM1 Antibody (Center) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | OPRM1 |
|---|---|
| Synonyms | MOR1 |
| Function | Receptor for endogenous opioids such as beta-endorphin and endomorphin (PubMed:12589820, PubMed:7891175, PubMed:7905839, PubMed:10529478, PubMed:7957926, PubMed:9689128). Receptor for natural and synthetic opioids including morphine, heroin, DAMGO, fentanyl, etorphine, buprenorphin and methadone (PubMed:12589820, PubMed:7891175, PubMed:7905839, PubMed:7957926, PubMed:10529478, PubMed:9689128, PubMed:10836142, PubMed:19300905). Also activated by enkephalin peptides, such as Met-enkephalin or Met-enkephalin-Arg-Phe, with higher affinity for Met-enkephalin-Arg-Phe (By similarity). Agonist binding to the receptor induces coupling to an inactive GDP-bound heterotrimeric G-protein complex and subsequent exchange of GDP for GTP in the G- protein alpha subunit leading to dissociation of the G-protein complex with the free GTP-bound G-protein alpha and the G-protein beta-gamma dimer activating downstream cellular effectors (PubMed:7905839). The agonist- and cell type-specific activity is predominantly coupled to pertussis toxin-sensitive G(i) and G(o) G alpha proteins, GNAI1, GNAI2, GNAI3 and GNAO1 isoforms Alpha-1 and Alpha-2, and to a lesser extent to pertussis toxin-insensitive G alpha proteins GNAZ and GNA15 (PubMed:12068084). They mediate an array of downstream cellular responses, including inhibition of adenylate cyclase activity and both N-type and L-type calcium channels, activation of inward rectifying potassium channels, mitogen-activated protein kinase (MAPK), phospholipase C (PLC), phosphoinositide/protein kinase (PKC), phosphoinositide 3-kinase (PI3K) and regulation of NF-kappa-B (By similarity). Also couples to adenylate cyclase stimulatory G alpha proteins (By similarity). The selective temporal coupling to G-proteins and subsequent signaling can be regulated by RGSZ proteins, such as RGS9, RGS17 and RGS4 (By similarity). Phosphorylation by members of the GPRK subfamily of Ser/Thr protein kinases and association with beta- arrestins is involved in short-term receptor desensitization (By similarity). Beta-arrestins associate with the GPRK-phosphorylated receptor and uncouple it from the G-protein thus terminating signal transduction (By similarity). The phosphorylated receptor is internalized through endocytosis via clathrin-coated pits which involves beta-arrestins (By similarity). The activation of the ERK pathway occurs either in a G-protein-dependent or a beta-arrestin- dependent manner and is regulated by agonist-specific receptor phosphorylation (By similarity). Acts as a class A G-protein coupled receptor (GPCR) which dissociates from beta-arrestin at or near the plasma membrane and undergoes rapid recycling (By similarity). Receptor down-regulation pathways are varying with the agonist and occur dependent or independent of G-protein coupling (By similarity). Endogenous ligands induce rapid desensitization, endocytosis and recycling (By similarity). Heterooligomerization with other GPCRs can modulate agonist binding, signaling and trafficking properties (By similarity). |
| Cellular Location | Cell membrane; Multi-pass membrane protein. Cell projection, axon {ECO:0000250|UniProtKB:P97266}. Perikaryon {ECO:0000250|UniProtKB:P97266}. Cell projection, dendrite {ECO:0000250|UniProtKB:P97266}. Endosome {ECO:0000250|UniProtKB:P97266}. Note=Is rapidly internalized after agonist binding. {ECO:0000250|UniProtKB:P97266} |
| Tissue Location | Expressed in brain. Isoform 16 and isoform 17 are detected in brain. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
OPRM1 is the mu opioid receptor, which is the primary site of action for the most commonly used opioids, including morphine, heroin, fentanyl, and methadone. It is also the primary receptor for endogenous opioid peptides beta-endorphin (see POMC, MIM 176830) and the enkephalins (see PENK, MIM 131330). The OPRM1 receptor is a membrane of the G protein-coupled receptor family (Bond et al., 1998 [PubMed 9689128]). There are at least 3 types of opioid receptors, mu, kappa (OPRK1; MIM 165196), and delta, each with a distinct pharmacologic profile.
References
Bare,L.A., et.al., FEBS Lett. 354 (2), 213-216 (1994)
Wang,J.B., et.al., FEBS Lett. 338 (2), 217-222 (1994)
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.