NALP1 Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IF, ICC, E |
|---|---|
| Primary Accession | Q9C000 |
| Other Accession | AAG30288, 11096303 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
| Calculated MW | 165866 Da |
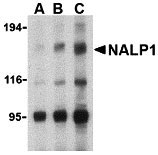
| Application Notes | NALP1 antibody can be used for detection of NALP1 by Western blot at 1 to 4 µg/mL. Antibody can also be used for immunocytochemistry starting at 10 µg/mL. For immunofluorescence start at 20 µg/mL. |
| Gene ID | 22861 |
|---|---|
| Other Names | NALP1 Antibody: NAC, CARD7, CIDED, NALP1, SLEV1, DEFCAP, PP1044, VAMAS1, CLR17.1, DEFCAP-L/S, KIAA0926, NAC, NACHT, LRR and PYD domains-containing protein 1, Caspase recruitment domain-containing protein 7, NLR family, pyrin domain containing 1 |
| Target/Specificity | NLRP1; |
| Reconstitution & Storage | NALP1 antibody can be stored at 4℃ for three months and -20℃, stable for up to one year. As with all antibodies care should be taken to avoid repeated freeze thaw cycles. Antibodies should not be exposed to prolonged high temperatures. |
| Precautions | NALP1 Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | NLRP1 {ECO:0000303|PubMed:22665479, ECO:0000312|HGNC:HGNC:14374} |
|---|---|
| Function | Acts as the sensor component of the NLRP1 inflammasome, which mediates inflammasome activation in response to various pathogen- associated signals, leading to subsequent pyroptosis (PubMed:22665479, PubMed:12191486, PubMed:17349957, PubMed:27662089, PubMed:31484767, PubMed:33093214, PubMed:33410748, PubMed:33731929, PubMed:33731932, PubMed:35857590). Inflammasomes are supramolecular complexes that assemble in the cytosol in response to pathogens and other damage- associated signals and play critical roles in innate immunity and inflammation (PubMed:22665479, PubMed:12191486, PubMed:17349957). Acts as a recognition receptor (PRR): recognizes specific pathogens and other damage-associated signals, such as cleavage by some human enteroviruses and rhinoviruses, double-stranded RNA, UV-B irradiation, or Val-boroPro inhibitor, and mediates the formation of the inflammasome polymeric complex composed of NLRP1, CASP1 and PYCARD/ASC (PubMed:22665479, PubMed:12191486, PubMed:17349957, PubMed:25562666, PubMed:30291141, PubMed:30096351, PubMed:33243852, PubMed:33093214, PubMed:33410748, PubMed:35857590). In response to pathogen-associated signals, the N-terminal part of NLRP1 is degraded by the proteasome, releasing the cleaved C-terminal part of the protein (NACHT, LRR and PYD domains-containing protein 1, C-terminus), which polymerizes and associates with PYCARD/ASC to initiate the formation of the inflammasome complex: the NLRP1 inflammasome recruits pro-caspase-1 (proCASP1) and promotes caspase-1 (CASP1) activation, which subsequently cleaves and activates inflammatory cytokines IL1B and IL18 and gasdermin-D (GSDMD), leading to pyroptosis (PubMed:22665479, PubMed:12191486, PubMed:17349957, PubMed:32051255, PubMed:33093214). In the absence of GSDMD expression, the NLRP1 inflammasome is able to recruit and activate CASP8, leading to activation of gasdermin-E (GSDME) (PubMed:33852854, PubMed:35594856). Activation of NLRP1 inflammasome is also required for HMGB1 secretion; the active cytokines and HMGB1 stimulate inflammatory responses (PubMed:22801494). Binds ATP and shows ATPase activity (PubMed:11113115, PubMed:15212762, PubMed:33243852). Plays an important role in antiviral immunity and inflammation in the human airway epithelium (PubMed:33093214). Specifically recognizes a number of pathogen-associated signals: upon infection by human rhinoviruses 14 and 16 (HRV-14 and HRV-16), NLRP1 is cleaved and activated which triggers NLRP1-dependent inflammasome activation and IL18 secretion (PubMed:33093214). Positive-strand RNA viruses, such as Semliki forest virus and long dsRNA activate the NLRP1 inflammasome, triggering IL1B release in a NLRP1-dependent fashion (PubMed:33243852). Acts as a direct sensor for long dsRNA and thus RNA virus infection (PubMed:33243852). May also be activated by muramyl dipeptide (MDP), a fragment of bacterial peptidoglycan, in a NOD2- dependent manner (PubMed:18511561). The NLRP1 inflammasome is also activated in response to UV-B irradiation causing ribosome collisions: ribosome collisions cause phosphorylation and activation of NLRP1 in a MAP3K20-dependent manner, leading to pyroptosis (PubMed:35857590). |
| Cellular Location | Cytoplasm, cytosol. Cytoplasm. Nucleus. Note=Nucleocytoplasmic distribution in lymphoid organs (probably in T-cells) and in neurons. In epithelial cells, predominantly cytoplasmic. [NACHT, LRR and PYD domains-containing protein 1, N-terminus]: Nucleus. Note=(Microbial infection) Interaction with human herpes virus 8/HHV-8 proteins ORF45 promotes translocation of the N-terminal part of NLRP1 into the nucleus, relieving autoinhibition of the NLRP1 inflammasome and leading to its activation. |
| Tissue Location | Widely expressed (PubMed:11113115, PubMed:17164409). Abundantly expressed in primary immune cells (isoform 1 and isoform 2), including in neutrophils, monocytes/macrophages, dendritic cells (mostly Langerhans cells), and B- and T-lymphocytes (at protein level) (PubMed:15285719, PubMed:17164409). Strongly expressed in epithelial cells lining the glandular epithelium, such as that of the gastrointestinal tract (stomach, small intestine, colon), the respiratory tract (trachea and bronchi), and the endometrial and endocervical glands, gallbladder, prostate, and breast (at protein level). In testis, expressed in spermatogonia and primary spermatocytes, but not in Sertoli cells (at protein level). In the brain, expressed in neurons, in particular in pyramidal ones and in oligodendrocytes, but not detected in microglia (at protein level) (PubMed:17164409). Expressed in adult and fetal ocular tissues, including in adult and 24-week old fetal choroid, sclera, cornea, and optic nerve, as well as in adult retina and fetal retina/retinal pigment epithelium (PubMed:23349227). Highly expressed in the skin throughout the epidermis and in dermal fibroblasts, in both glabrous skin and plantar skin. It is detected in keratinocytes, but not in melanocytes. Expressed in epidermal appendages such as hair follicles (PubMed:27662089). |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
NALP1 Antibody: NALP1 belongs to a family of cytoplasmic proteins that have been implicated in cell responses to apoptotic and inflammatory stimuli. It contains a caspase recruitment domain (CARD) in addition to a NACHT domain and leucine rich repeat (LRR). This protein interacts strongly with caspase 2 and weakly with caspase 9, as well as with Apaf-1. Transient overexpression of this gene in cultured cells was sufficient to induce apoptosis. NALP1 mRNA and protein expression levels in myeloid leukemia cells are increased following CREB (cAMP-response element binding protein) activation, suggesting that NALP1 may contribute to modulate the response of these cells to pro-inflammatory stimuli. Multiple alternatively spliced transcript variants encoding distinct isoforms have been found for NALP1.
References
NALP1 antibody can be stored at 4˚C, stable for one year. Tschopp J, Martinon F, and Burns K. NALPs: a novel protein family involved in inflammation. Nat. Rev. Mol. Cell Biol. 2003; 4:95-104.
Chu ZL, Pio F, Xie Z, et al. A novel enhancer of the Apaf1 apoptosome involved in cytochrome c-dependent caspase activation and apoptosis. J. Biol. Chem. 2001; 276:9239-45.
Hlaing T, Guo RF, Dilley KA, et al. Molecular cloning and characterization of DEFCAP-L and -S, two isoforms of a novel member of the Ced-4 family of apoptosis proteins. J. Biol. Chem. 2001; 276:9230-8.
Sanz C, Calasanz MJ, Andreu E, et al. NALP1 is a transcriptional target for cAMP-response-element-binding protein (CREB) in myeloid leukemia cells. Biochem. J. 2004; 384:281-6.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.