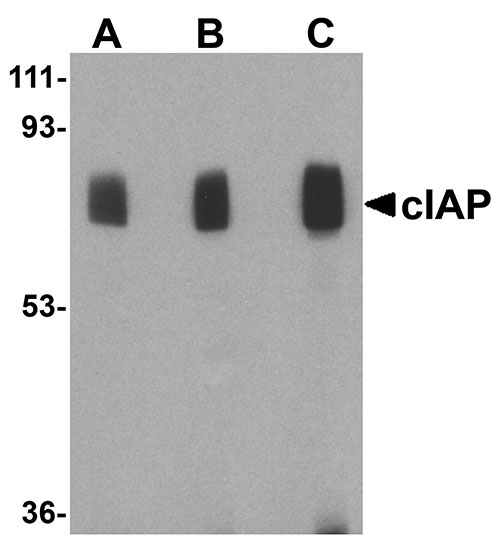
cIAP Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC-P, IF, E |
|---|---|
| Primary Accession | Q13490 |
| Other Accession | NP_001157, 4502141 |
| Reactivity | Human, Mouse |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
| Calculated MW | 69900 Da |
| Application Notes | cIAP antibody can be used for the detection of c-IAP by Western blot at 1 to 4 µg/mL. Antibody can also be used for immunohistochemistry starting at 10 µg/mL. For immunofluorescence start at 20 µg/mL. |
| Gene ID | 329 |
|---|---|
| Other Names | cIAP Antibody: API1, MIHB, HIAP2, RNF48, cIAP1, Hiap-2, c-IAP1, API1, IAP2, Baculoviral IAP repeat-containing protein 2, C-IAP1, IAP-2, baculoviral IAP repeat-containing 2 |
| Target/Specificity | BIRC2; |
| Reconstitution & Storage | cIAP antibody can be stored at 4℃ for three months and -20℃, stable for up to one year. As with all antibodies care should be taken to avoid repeated freeze thaw cycles. Antibodies should not be exposed to prolonged high temperatures. |
| Precautions | cIAP Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | BIRC2 |
|---|---|
| Synonyms | API1, MIHB, RNF48 |
| Function | Multi-functional protein which regulates not only caspases and apoptosis, but also modulates inflammatory signaling and immunity, mitogenic kinase signaling, and cell proliferation, as well as cell invasion and metastasis. Acts as an E3 ubiquitin-protein ligase regulating NF-kappa-B signaling and regulates both canonical and non- canonical NF-kappa-B signaling by acting in opposite directions: acts as a positive regulator of the canonical pathway and suppresses constitutive activation of non-canonical NF-kappa-B signaling. The target proteins for its E3 ubiquitin-protein ligase activity include: RIPK1, RIPK2, RIPK3, RIPK4, CASP3, CASP7, CASP8, TRAF2, DIABLO/SMAC, MAP3K14/NIK, MAP3K5/ASK1, IKBKG/NEMO, IKBKE and MXD1/MAD1. Can also function as an E3 ubiquitin-protein ligase of the NEDD8 conjugation pathway, targeting effector caspases for neddylation and inactivation. Acts as an important regulator of innate immune signaling via regulation of Toll-like receptors (TLRs), Nodlike receptors (NLRs) and RIG-I like receptors (RLRs), collectively referred to as pattern recognition receptors (PRRs). Protects cells from spontaneous formation of the ripoptosome, a large multi-protein complex that has the capability to kill cancer cells in a caspase-dependent and caspase- independent manner. Suppresses ripoptosome formation by ubiquitinating RIPK1 and CASP8. Can stimulate the transcriptional activity of E2F1. Plays a role in the modulation of the cell cycle. |
| Cellular Location | Cytoplasm. Nucleus. Note=Agents that induce either the extrinsic or intrinsic apoptotic pathways promote its redistribution from the nuclear compartment to the cytoplasmic compartment. Associated with the midbody in telophase cells, and found diffusely in the nucleus of interphase cells |
| Tissue Location | Present in many fetal and adult tissues. Mainly expressed in adult skeletal muscle, thymus, testis, ovary, and pancreas, low or absent in brain and peripheral blood leukocytes |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
cIAP Antibody: Apoptosis, or programmed cell death, is related to many diseases, such as cancer. Apoptosis is triggered by a variety of stimuli including members in the TNF family and can be prevented by the inhibitor of apoptosis (IAP) proteins. IAP proteins form a conserved gene family that binds to and inhibits cell death proteases. The two isoforms of c-IAP (c-IAP1 and c-IAP2) are structurally related to XIAP, containing 3 baculoviral IAP repeat (BIR) motifs that are essential and sufficient for the binding and inhibition of caspases-3, -7. The c-IAPs can associate with the death receptor TNF-R2, and mediate the ubiquitinization of TRAF2 following the binding of TNF-α by its receptor. Omi, a negative regulator of c-IAP, inhibits its activity by catalytically cleaving c-IAP. Another negative regulator, Smac/DIABLO, acts by enhancing the auto-ubiquitization activity of c-IAP.
References
Schimmer AD. Inhibitor of apoptosis proteins: translating basic knowledge into clinical practice. Cancer Res. 2004; 64:7183-90.
Rothe M, Pan M-G, Henzel WJ, et al. The TNFR2-TRAF signaling complex contains two novel proteins related to baculoviral inhibitor of apoptosis proteins. Cell 1995; 83:1243-52.
Deveraux QL, Leo E, Stennicke HR, et al. IAPs block apoptotic events induced by caspase-8 and cytochrome c by direct inhibition of distinct caspases. EMBO J. 1998; 17:2215-23.
Li X, Yang Y, Ashwell JD. TNF-RII and c-IAP1 mediate ubiquitinization and degradation of TRAF 2. Nature 2002; 416:345-7.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.