ERAP1 Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

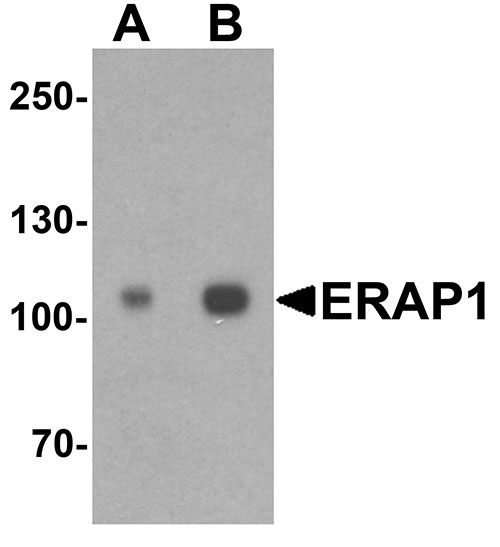
Application
| WB, IHC-P, IF, E |
|---|---|
| Primary Accession | Q9NZ08 |
| Other Accession | NP_057526, 94818901 |
| Reactivity | Human, Mouse |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
| Calculated MW | Predicted: 104 kDa Observed: 105kDa |
| Application Notes | ERAP1 antibody can be used for detection of ERAP1 by Western blot at 1 - 2 µg/ml. |
| Gene ID | 51752 |
|---|---|
| Target/Specificity | ERAP1; ERAP1 antibody is human and mouse reactive. At least two isoforms of ERAP1 are known to exist; this antibody will detect both isoforms. ERAP1 antibody is predicted to not cross-react with ERAP2. |
| Reconstitution & Storage | ERAP1 antibody can be stored at 4℃ for three months and -20℃, stable for up to one year. |
| Precautions | ERAP1 Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | ERAP1 |
|---|---|
| Synonyms | APPILS, ARTS1, KIAA0525 |
| Function | Aminopeptidase that plays a central role in peptide trimming, a step required for the generation of most HLA class I-binding peptides. Peptide trimming is essential to customize longer precursor peptides to fit them to the correct length required for presentation on MHC class I molecules. Strongly prefers substrates 9-16 residues long. Rapidly degrades 13-mer to a 9-mer and then stops. Preferentially hydrolyzes the residue Leu and peptides with a hydrophobic C-terminus, while it has weak activity toward peptides with charged C-terminus. May play a role in the inactivation of peptide hormones. May be involved in the regulation of blood pressure through the inactivation of angiotensin II and/or the generation of bradykinin in the kidney. |
| Cellular Location | Endoplasmic reticulum membrane; Single-pass type II membrane protein |
| Tissue Location | Ubiquitous. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
The endoplasmic reticulum (ER) aminopeptidase 1 (ERAP1), a member of the peptidase M1 family, plays a central role in peptide trimming, a step required for the generation of most HLA class I-binding peptides (1,2). It is also designated as adipocyte-derived leucine aminopeptidase (A-LAP), puromycin-insensitive leucine-specific aminopeptidase (PILS-AP) and aminopeptidase regulator of TNFR1 shedding (ARTS-1) (3). ERAP1 is localized to the lumen of the ER and induced by interferon. It may be involved in the regulation of blood pressure through the inactivation of angiotensin II and/or the generation of bradykinin in the kidney (3,4).
References
Hattori A, Kitatani K, Matsumoto H, et al. Characterization of recombinant human adipocytederived leucine aminopeptidase expressed in Chinese hamster ovary cells. J. Biochem. 2000; 128:755-62.
Saric T, Chang SC, Hattori A, et al. An IFN-gamma induced aminopeptidase in the ER, ERAP1, trims precursors to MHC class I-presented peptides. Nat. Immunol. 2002; 3:1169-76.
Cui X, Hawari F, Alsaaty S, et al. Identification of ARTS-1 as a novel TNFR1-binding protein that promotes TNFR1 ectodomain shedding. J. Clin. Invest. 2002; 110:515-26.
Akada T, Yamazaki T, Miyashita H, et al. Puromycin insensitive leucyl-specific aminopeptidase (PILSAP) is involved in the activation of endothelial integrins. J. Cell Physiol. 2002; 193: 253-62.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.













 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.