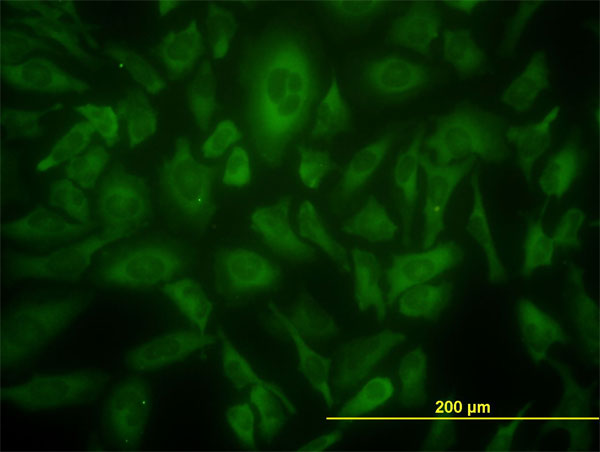
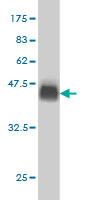
USP9X Antibody (monoclonal) (M01)
Mouse monoclonal antibody raised against a partial recombinant USP9X.
- SPECIFICATION
- CITATIONS: 1
- PROTOCOLS
- BACKGROUND

Application
| WB, IF, E |
|---|---|
| Primary Accession | Q93008 |
| Other Accession | NM_021906 |
| Reactivity | Human |
| Host | Mouse |
| Clonality | Monoclonal |
| Isotype | IgG1 Kappa |
| Clone Names | 1C4 |
| Calculated MW | 290463 Da |
| Gene ID | 8239 |
|---|---|
| Other Names | Probable ubiquitin carboxyl-terminal hydrolase FAF-X, Deubiquitinating enzyme FAF-X, Fat facets in mammals, hFAM, Fat facets protein-related, X-linked, Ubiquitin thioesterase FAF-X, Ubiquitin-specific protease 9, X chromosome, Ubiquitin-specific-processing protease FAF-X, USP9X, DFFRX, FAM, USP9 |
| Target/Specificity | USP9X (NP_068706, 1 a.a. ~ 90 a.a) partial recombinant protein with GST tag. MW of the GST tag alone is 26 KDa. |
| Dilution | WB~~1:500~1000 |
| Format | Clear, colorless solution in phosphate buffered saline, pH 7.2 . |
| Storage | Store at -20°C or lower. Aliquot to avoid repeated freezing and thawing. |
| Precautions | USP9X Antibody (monoclonal) (M01) is for research use only and not for use in diagnostic or therapeutic procedures. |

Provided below are standard protocols that you may find useful for product applications.
Background
This gene is a member of the peptidase C19 family and encodes a protein that is similar to ubiquitin-specific proteases. Though this gene is located on the X chromosome, it escapes X-inactivation. Mutations in this gene have been associated with Turner syndrome. Alternate transcriptional splice variants, encoding different isoforms, have been characterized. [provided by RefSeq]
References
1.Role of Ku70 in deubiquitination of Mcl-1 and suppression of apoptosis.Wang B, Xie M, Li R, Owonikoko TK, Ramalingam SS, Khuri FR, Curran WJ, Wang Y, Deng XCell Death Differ. 2014 Apr 25. doi: 10.1038/cdd.2014.42.2.Deubiquitinase USP9x Confers Radioresistance through Stabilization of Mcl-1.Trivigno D, Essmann F, Huber SM, Rudner J.Neoplasia. 2012 Oct;14(10):893-904.3.Mcl-1 phosphorylation defines ABT-737 resistance that can be overcome by increased NOXA expression in leukemic B-cells.Mazumder S, Choudhary GS, Al-Harbi S, Almasan A.Cancer Res. 2012 Apr 23.4.The Bcl-xL inhibitor, ABT-737, efficiently induces apoptosis and suppresses growth of hepatoma cells in combination with sorafenib.Hikita H, Takehara T, Shimizu S, Kodama T, Shigekawa M, Iwase K, Hosui A, Miyagi T, Tatsumi T, Ishida H, Li W, Kanto T, Hiramatsu N, Hayashi N.Hepatology (2010) DOI: 10.1002/ hep.23836
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.














 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.